THE UNITED STATES SECURITIES AND EXCHANGE COMMISSION
(“SEC”) DOES NOT PASS UPON THE MERITS OF OR GIVE ITS APPROVAL TO ANY SECURITIES OFFERED OR THE TERMS OF THE OFFERING, NOR
DOES IT PASS UPON THE ACCURACY OR COMPLETENESS OF ANY OFFERING CIRCULAR OR OTHER SELLING LITERATURE. THESE SECURITIES ARE OFFERED PURSUANT
TO AN EXEMPTION FROM REGISTRATION WITH THE COMMISSION; HOWEVER, THE COMMISSION HAS NOT MADE AN INDEPENDENT DETERMINATION THAT THIS INVESTMENT
INVOLVES A DEGREE OF RISK THAT MAY NOT BE SUITABLE FOR ALL PERSONS. ONLY THOSE INVESTORS WHO CAN BEAR THE LOSS OF A SIGNIFICANT PORTION
OF THEIR INVESTMENT SHOULD PARTICIPATE IN THE INVESTMENT. (SEE “RISK FACTORS” BELOW.)
AN OFFERING STATEMENT PURSUANT TO REGULATION A RELATING
TO THESE SECURITIES HAS BEEN FILED WITH THE SECURITIES AND EXCHANGE COMMISSION. INFORMATION CONTAINED IN THIS PRELIMINARY OFFERING CIRCULAR
IS SUBJECT TO COMPLETION OR AMENDMENT. THESE SECURITIES MAY NOT BE SOLD NOR MAY OFFERS TO BUY BE ACCEPTED BEFORE THE OFFERING STATEMENT
FILED WITH THE COMMISSION IS QUALIFIED. THIS PRELIMINARY OFFERING CIRCULAR SHALL NOT CONSTITUTE AN OFFER TO SELL OR THE SOLICITATION OF
AN OFFER TO BUY NOR MAY THERE BE ANY SALES OF THESE SECURITIES IN ANY STATE IN WHICH SUCH OFFER, SOLICITATION OR SALE WOULD BE UNLAWFUL
BEFORE REGISTRATION OR QUALIFICATION UNDER THE LAWS OF ANY SUCH STATE. THE COMPANY MAY ELECT TO SATISFY ITS OBLIGATION TO DELIVER A FINAL
OFFERING CIRCULAR BY SENDING YOU A NOTICE WITHIN TWO BUSINESS DAYS AFTER THE COMPLETION OF THE SALE THAT CONTAINS THE URL WHERE THE FINAL
OFFERING CIRCULAR OR THE OFFERING STATEMENT IN WHICH SUCH FINAL OFFERING CIRCULAR WAS FILED MAY BE OBTAINED.
THE SECURITIES OFFERED HAVE NOT BEEN APPROVED OR DISAPPROVED
BY ANY STATE REGULATORY AUTHORITY NOR HAS ANY STATE REGULATORY AUTHORITY PASSED UPON OR ENDORSED THE MERITS OF THE OFFERING OR THE ACCURACY
OR ADEQUACY OF THIS OFFERING CIRCULAR. ANY REPRESENTATION TO THE CONTRARY IS UNLAWFUL.
Preliminary Offering Circular
For
Kurve Therapeutics, Inc.
A Delaware Corporation
May 20, 2022
SECURITIES OFFERED : Equity in the form
of Shares of common stock
PRICE PER SHARE : $5.80 per Share of Common
Stock
MAXIMUM OFFERING AMOUNT : $50,000,000.00
MINIMUM OFFERING AMOUNT : Not Applicable (No Minimum Offering Amount)
MINIMUM INVESTMENT : $580.00
CONTACT INFORMATION :
Kurve Therapeutics, Inc.
16825 48th Ave West, Suite 434
Lynnwood, WA 98037
(425) 640-9249
Generally, no sale may be made to you in this Offering
if the aggregate purchase price you pay is more than ten (10%) percent of the greater of your annual income or net worth. Different rules
apply to accredited investors and non-natural persons. Before making any representation that your investment does not exceed applicable
thresholds, Investors are encouraged to review rule 251(d)(2)(i)(C) of Regulation A. For general information on investing, Investors are
encouraged to refer to www.investor.gov.
This Offering Circular conforms
to SEC Form 1-A.
Kurve Therapeutics, Inc. (the “Company”
or “Kurve” or the “Issuer”) is a Delaware corporation, originally formed on November 21, 2019 by filing Articles
of Incorporation with the Secretary of the State of Delaware. The Company registered as a foreign corporation with the Washington Secretary
of State on December 3, 2019. (see Exhibit 2 “Articles of Incorporation and Certificate of Registration”). The Company is
offering (the “Offering”) by means of this offering circular (the “Offering Circular”) Company equity in the form
of common stock denominated in shares (the “Shares”) on a “best efforts” and ongoing basis to investors who meet
the Investor Suitability standards as set forth herein. (See “Investor Suitability” below.) The Company will offer Shares
through kurvetx.com (“Platform”), and through Rialto Markets, a FINRA-registered broker-dealer entitled to commissions upon
the sale of the Shares. (See the “Terms of the Offering” below.) Persons who purchase Shares will be shareholders of the Company
(“Shareholders” or “Investors”). The Company has two main products. The first product is Intrulinâ,
Kurve intranasal insulin, which is a drug the Company is developing to combat cognitive aging, neurodegenerative diseases and related
disorders. The second product is the Company’s patented Controlled Particle Dispersion® (“CPD”) technology, an intranasal
delivery system. ViaNaseâ is the brand name for the product/device that utilizes the CPD
technology. For purposes of this Offering Circular, the Intrulin and ViaNase will be referred herein as the “Products” –
however, these are more accurately referred to as “product candidates” since both are still subject to FDA approval/authorization
and have not been cleared for commercial use. See “Risk Factors” and “Description of the Business” below.
The Company intends to use the Proceeds of this
Offering to (1) Complete the purchase of select intellectual property from Kurve Technology, Inc. (through Savile Therapeutics,
Inc.).; (2) Design, develop, and execute human clinical trials for the Company’s Products; (3) Develop relationships with
select producers of CNS drugs for potential licensing relationships; (4) Expand research and development activities for the
Company’s Products; (5) Gain FDA Approval/authorization for the Company’s Products; (6) Marketing the Offering and the
Company’s Products; (7) Selling Shareholders.
The minimum investment amount per Investor is Five
Hundred Eighty dollars ($580.00), representing One Hundred (100) Shares at Five Dollars and Eighty Cents ($5.80) per Share. Although the
Company does not intend to list the Shares for trading on any exchange or other trading market.
The Company is a run by a board of directors, comprised
of two (2) directors (the “Board” collectively, “Director” when referring to a director). The day-to-day management
and investment decisions of the Company are vested in the Board and in the Officer.
Sales of the Shares pursuant to the Offering will
commence immediately upon qualification of the Offering by the Securities and Exchange Commission (the “Effective Date”) and
will terminate at the discretion of the Board or twelve (12) months following the Effective Date, whichever is earlier. The maximum amount
of the Offering shall not exceed Fifty Million Dollars ($50,000,000) in any twelve (12) month period (“Maximum Offering Amount”)
in accordance with Tier II of Regulation A as set forth under the Securities Act of 1933, as , (“Reg A Tier II” or “Tier
II”). The Company intends to offer the Shares described herein on a continuous and ongoing basis pursuant to Rule 251(d)(3)(i)(f).
Further, the acceptance of Investor subscriptions, may be briefly paused at times to allow the Company to effectively and accurately process
and settle subscriptions that have been received. (See “Terms of the Offering” below.) The Company reserves the right to increase
the Maximum Offering Amount at any time before the termination of the Offering, subject to qualification by the SEC of a post-qualification
amendment.
Prior to this Offering, there has been no public market
for the Shares, and none is expected to develop. The Offering price is arbitrary and does not bear any relationship to the value of the
assets of the Company. The Company does not currently have plans to list any Shares on any securities market, however it reserves the
right to do so in the future. Investing in the Company through the purchase of Shares involves risks, some of which are set forth below.
See the section titled “Risk Factors” to read about the factors an Investor should consider prior to purchasing Shares.
Investors who purchase Shares will become Shareholders
of the Company (“Investors” or “Shareholders” subject to the terms of the Amended and Restated Certificate of
Incorporation (the “Certificate of Incorporation”) and the Bylaws of the Company (see Exhibit 2A, the “Certificate of
Incorporation” and 2B “Bylaws”) once the Company deposits the Investor’s investment into the Company’s main
operating account.
The Directors and Officers will receive
compensation from the Company as employees. (See “Risk Factors”, “Compensation of Directors and Officers”
below.) Investing in the Shares is speculative and involves substantial risks, including risk of complete loss. Prospective
Investors should purchase these securities only if they can afford a complete loss of their investment. (See “Risk
Factors” below starting on Page 4.)
As of the date of this Offering Circular, the Company
has engaged KoreConX as transfer agent in relation to this Offering.
RULE 251(D)(3)(I)(F) DISCLOSURE. RULE 251(D)(3)(I)((F)
PERMITS REGULATION A OFFERINGS TO CONDUCT ONGOING CONTINUOUS OFFERINGS OF SECURITIES FOR MORE THAN THIRTY (30) DAYS AFTER THE QUALIFICATION
DATE IF: (1) THE OFFERING WILL COMMENCE WITHIN TWO (2) DAYS AFTER THE QUALIFICATION DATE; (2) THE OFFERING WILL BE MADE ON A CONTINUOUS
AND ONGOING BASIS FOR A PERIOD THAT MAY BE IN EXCESS OF THIRTY (30) DAYS OF THE INITIAL QUALIFICATION DATE; (3) THE OFFERING WILL BE IN
AN AMOUNT THAT, AT THE TIME THE OFFERING CIRCULAR IS QUALIFIED, IS REASONABLY EXPECTED TO BE OFFERED AND SOLD WITHIN THREE (3) YEARS FROM
THE INITIAL QUALIFICATION DATE; AND (4) THE SECURITIES MAY BE OFFERED AND SOLD ONLY IF NOT MORE THAN THREE (3) YEARS HAVE ELAPSED SINCE
THE INITIAL QUALIFICATION DATE OF THE OFFERING, UNLESS A NEW OFFERING CIRCULAR IS SUBMITTED AND FILED BY THE COMPANY PURSUANT TO RULE
251(D) (3)(I)((F) WITH THE SEC COVERING THE REMAINING SECURITIES OFFERED UNDER THE PREVIOUS OFFERING; THEN THE SECURITIES MAY CONTINUE
TO BE OFFERED AND SOLD UNTIL THE EARLIER OF THE QUALIFICATION DATE OF THE NEW OFFERING CIRCULAR OR THE ONE HUNDRED EIGHTY (180) CALENDAR
DAYS AFTER THE THIRD ANNIVERSARY OF THE INITIAL QUALIFICATION DATE OF THE PRIOR OFFERING CIRCULAR. THE COMPANY INTENDS TO OFFER THE SHARES
DESCRIBED HEREIN ON A CONTINUOUS AND ONGOING BASIS PURSUANT TO RULE 251(D)(3)(I)(F). THE COMPANY INTENDS TO COMMENCE THE OFFERING IMMEDIATELY
AND NO LATER THAN TWO (2) DAYS FROM THE INITIAL QUALIFICATION DATE. THE COMPANY REASONABLY EXPECTS TO OFFER AND SELL THE SECURITIES STATED
IN THIS OFFERING CIRCULAR WITHIN ONE (1) YEAR FROM THE INITIAL QUALIFICATION DATE.
The Company will commence sales of the Shares immediately
upon qualification of the Offering by the SEC. The Company approximates sales will commence during Q2 - 2022
| | |
Price to Public* | |
Commissions** | |
Proceeds to Other Persons*** | |
Proceeds to the Company |
| Amount to be Raised per Share | |
$ | 5.80 | | |
$ | 0.174 | | |
$ | 0.879 | | |
$ | 5.626 | |
| Minimum Investment Amount | |
$ | 580 | | |
$ | 17.40 | | |
$ | 87.90 | | |
$ | 562.60 | |
| Minimum Offering Amount | |
| N/A | | |
| N/A | | |
| N/A | | |
| N/A | |
| Maximum Offering Amount | |
$ | 50,000,000 | | |
$ | 1,500,000 | | |
| 7,579,816.88 | | |
$ | 40,920,183.11 | |
*The Offering price to Investors
was arbitrarily determined by the Board.
** The Company is not using an underwriter
for the sale of Shares. These commissions listed are those for Rialto Markets, a FINRA broker-dealer. Rialto Markets is entitled to 3%
on all sales of securities as placement agent. If securities are sold through the efforts of Rialto Markets an additional 8% will be due
to Rialto Markets up to a maximum of $800,000. The commissions due to Rialto Markets are conditional on the services provided by Rialto
Markets with respect to any one sale. See “Plan of Distribution “ below.
***These proceeds are directed to selling securityholders.
See “Plan of Distribution and Selling Securityholders” below.
FORWARD LOOKING STATEMENTS
Investors should not rely on forward-looking statements
because they are inherently uncertain. Investors should not rely on forward-looking statements in this Offering Circular. This Offering
Circular contains forward-looking statements that involve risks and uncertainties. The use of words such as “anticipated”,
“projected”, “forecasted”, “estimated”, “prospective”, “believes”, “expects,”
“plans”, “future”, “intends”, “should”, “can”, “could”, “might”,
“potential”, “continue”, “may”, “will”, and similar expressions identify these forward-looking
statements. Investors should not place undue reliance on these forward-looking statements, which may apply only as of the date of this
Offering Circular.
TABLE OF CONTENTS
SUMMARY OF THE OFFERING
The following information is only a brief summary
of, and is qualified in its entirety by, the detailed information appearing elsewhere in this Offering. This Offering Circular, together
with the exhibits attached including, but not limited to, the Certificate of Incorporation and Company Bylaws, copies of which are attached
hereto as Exhibit 2A and Exhibit 2B, respectively, and should be carefully read in their entirety before any investment decision is made.
If there is a conflict between the terms contained in this Offering Circular and these documents, Certificate of Incorporation and Bylaws
shall prevail and control, and no Investor should rely on any reference herein to the Certificate of Incorporation or Bylaws without consulting
the actual underlying documents.
| COMPANY INFORMATION AND BUSINESS |
Kurve Therapeutics, Inc. is a Delaware corporation with a principal place of business located at Kurve Therapeutics, Inc. 16825 48th Ave West, Suite 434, Lynnwood, WA 98037. |
| MANAGEMENT |
The Company is managed by a Board of Directors. The Board is comprised of two (2) Directors, Marc Giroux and Thomas McDowell, who also hold the titles of Chief Executive Officer and Chief Financial Officer of the Company, respectively. |
| THE OFFERING |
This Offering is the first capital raise by the Company in its history. The Company is selling Company equity in the form of Common Stock (the “Common Stock” or the “Shares”) through this Offering. The Company will use the Proceeds of this Offering to commence in additional clinical trials and scale up its operations. See “Use of Proceeds” below. |
| SECURITIES BEING OFFERED |
The Shares are being offered at a purchase price of $5.80 per Share. The Minimum Offering Amount for any Investor is $580.00 or 100 Shares. Upon purchase of the Shares, a Shareholder is granted (i) the right to vote on all matters subject to a Common Stock vote; and (ii) the right to receive dividends or disbursements, when the Board declares such dividends or disbursements. For a complete summary of the rights granted to Shareholders, see “Description of the Securities” below. |
| COMPENSATION TO DIRECTORS AND OFFICERS |
The Company pays its Directors and Officers salaries for their roles as Directors and Officers. For more information on his compensation, see “Compensation of the Directors and Officers” section below. The Directors, Officers, and employees of the Company will not be compensated through commissions for the sale of the Shares through this Offering. |
| PRIOR EXPERIENCE OF COMPANY MANAGEMENT |
The Company’s officers and Directors have extensive experience in life sciences, medical devices, and pharmaceuticals industries. For more information Please see “Directors and Officers” below. |
| INVESTOR SUITABILITY STANDARDS |
The Shares will not be sold to any person unless they
are a “Qualified Purchaser”. A Qualified Purchaser includes: (1) an “Accredited Investor” as that term is defined
in Rule 501(a) of Regulation D promulgated under the Securities Act of 1933 (the “Securities Act”); or (2) all other Investors
who meet the investment limitations set forth in Rule 251(d)(2)(C) of Regulation A. Such persons as stated in (2) above must conform with
the “Limitations on Investment Amount” as described in the next below.
Each person purchasing Shares will be subject to the
terms of the Certificate of Incorporation and Bylaws, a copy of which attached hereto as Exhibit 2A and Exhibit 2B, respectively.
Each person acquiring Shares may be required to represent
that he, she, or it is purchasing the Shares for his, her, or its own account for investment purposes and not with a view to resell or
distribute the securities.
Each prospective purchaser of Shares may be required
to furnish such information or certification as the Company may require determining whether any person or entity purchasing Shares is
an Accredited Investor if such is claimed by the Investor. |
| LIMITATIONS ON INVESTMENT AMOUNT |
For Qualified Purchasers who are Accredited Investors,
there is no limitation as to the amount invested through the purchase of Shares. For non-Accredited Investors, the aggregate purchase
price paid to the Company for the purchase of the Shares cannot be more than 10% of the greater of the purchaser’s (1) annual income
or net worth if purchaser is a natural person; or (2) revenue or net assets for the purchaser’s most recently completed fiscal year
if purchaser is a non-natural person.
Different rules apply to Accredited Investors and
non-natural persons. Each Investor should review Rule 251(d)(2)(i)(C) of Regulation A before purchasing the Shares. |
| COMMISSIONS FOR SELLING Shares |
The Shares will be offered and sold directly by the
Company, the Board, the Officers, and Company’s employees. No commissions for selling the Shares will be paid to the Company, the
Board, the Officer, or the Company’s employees.
The Company has engaged Rialto Markets, a registered
independent FINRA broker-dealer, as placement agent for this Offering. Rialto Markets is entitled to 3% on all sales of securities as
placement agent. If securities are sold through the efforts of Rialto Markets an additional 8% will be due to Rialto Markets up to a maximum
of $1,600,000. The commissions due to Rialto Markets are conditional on the services provided by Rialto Markets with respect to any one
sale. See “Plan of Distribution “ below.
|
| NO LIQUIDITY |
There is no public market for the Shares, and none
is expected to develop in the near future. Additionally, the Shares will be transferable, in accordance with Delaware law. However, the
shares will not be listed for trading on any exchange or automated quotation system. (See “Description of the Securities”
below.) Prospective Investors are urged to consult their own legal advisors with respect to secondary trading of the Shares. (See “Risk
Factors” below.)
|
| SELLING SECURITYHOLDERS |
The Company intends on having Selling Shareholders as part of this Offering. 1,306,862 Shares are being offered on the account of Selling Shareholders through this offering. This equals $7,579,816.88 of the Proceeds. See “Plan of Distribution and Selling Shareholders” below. |
| BONUS SHARES |
The Company intends to issue bonus shares (“Bonus Shares”) to Investors who invest in the Offering. For more information, please see “Plan of Distribution” below. |
| COMPANY EXPENSES |
Except as otherwise provided herein, the Company shall bear all costs and expenses associated with the Offering, the operation of the Company, including, but not limited to, the annual tax preparation of the Company's tax returns, any state and federal income tax due, accounting fees, filing fees, independent audit reports, costs and expenses associated with the acquisition, rehabilitation, holding and management of real estate and costs and expenses associated with the disposition of real estate or other Company assets. |
RISK FACTORS
The SEC requires the Company to identify risks
that are specific to its business and its financial condition. The Company is still subject to all the same risks that all companies
in its business, and all companies in the economy, are exposed to. These include risks relating to economic downturns, political and
economic events and technological developments (such as hacking and the ability to prevent hacking). Additionally, early-stage
companies are inherently riskier than more developed companies. You should consider general risks as well as specific risks when
deciding whether to invest.
RISKS RELATED TO THE COMPANY
AND ITS BUSINESS
The Company is has limited operating history.
The Company has limited established business operations,
and it is unclear at this point which, if any, of the Company’s current and intended plans may come into fruition and, if they do,
which ones will be a success. The Company has incurred a net loss and has recorded limited revenues since inception. There is no assurance
that the Company will ever be able to establish successful business operations, become profitable or generate sufficient revenues to operate
business or pay dividends.
The loss of any member of the Company’s management team or
their inability to attract and retain highly skilled scientists, engineers, clinicians, and salespeople could adversely affect company
business.
Company success depends on the skills, experience
and performance of key members of the Company’s management team, including the Officers and Directors. The individual and collective
efforts of the Officers and Directors will be essential as the Company continues to develop the Company and Products.
Company research and development and manufacturing
operations depend on the Company’s ability to attract and retain highly-skilled employees including scientists, technicians and
engineers. The Company may not be able to attract or retain a sufficient number of qualified highly-skilled employees in the future due
to the competition for qualified personnel in the pharmaceutical/medical device industry. The Company also faces competition from universities
and public and private research institutions in recruiting and retaining highly qualified scientific personnel. Recruiting and retention
difficulties can limit the ability to support Company. The Company also does not maintain “key person” insurance on any of
Company employees.
The Company may not be able to develop commercially viable products
on the anticipated timetable, or at all.
The Company’s products, namely the CPD
technology and Intrulin (the “Products”), may be difficult to scale to a commercially viable level since it must meet
expectations that it is equivalent or superior to the existing products in terms of efficacy, safety, and cost efficiency. The
Company still needs to develop and refine the Products and the manufacturing process for its Products to ensure that the Products
meets performance goals, cost targets, and regulatory approval. The Company needs to perform additional laboratory and clinical
trials and may encounter problems and delays. If the clinical trials reveal technical defects or reveal that the Company’s
Products do not meet performance goals and cost targets, the Company’s commercialization schedule could be delayed as the
Company attempts to devise solutions to the defects or problems. If the Company is unable to find solutions, the Company’s
business may not be viable.
The Company may not be able to successfully execute the business
plan.
In addition to the requirement to successfully develop
the Products for commercial success, the Company must also raise significant amounts of capital, foster relationships with key suppliers
and attract customers. There is no guarantee that the Company will be able to achieve or sustain any of the foregoing within the anticipated
timeframe or at all. The Company may exceed the budget, encounter obstacles in research and development activities, or be hindered or
delayed in implementing the Company’s commercialization plans, any of which could imperil the Company’s ability to secure
customer contracts and begin generating revenues. In addition, any such delays or problems would require the Company to secure additional
funding over and above what the Company currently anticipates they require to sustain business, which the Company may not be able to raise.
The Company has only two main product lines.
The Company’s primary products are the Intrulin
and the CPD technology. Even though these are two distinct products, with disparate regulatory and design/manufacturing paths –
the CPD technology is designed to deliver the Intrulin (and other Company products). The Company may sell of license the CPD technology
as a delivery device for third party-developed drugs. The Company’s survival in near term depends on being able to sell these Products
to a sufficient number of customers to make a profit. The Company’s current customer base is still small, and the Company will only
succeed if it can attract more customers for its primary product and maintain those customers.
The Company has not yet generated significant revenues or profits
and it may take a long time for the Company to become profitable.
The Company and has not yet generated any significant
revenue or profit and no revenue or profit from the sale of the Products. The Company does not yet have customers, since the gaining of
customers is conditional upon regulatory approval/authorization of the Intrulin and CPD technology (see “Description of the Business”
below). The Company hopes to obtain approval/authorization of the Products by the U.S. Food and Drug Administration (“FDA”)
and other international regulatory agencies. The Company is working towards having sales of the Products, but the Company anticipates
that it will take 48 months to reach a breakeven point and at least fifty-four (54) months achieve profitability, if at all.
The Company’s ability to raise capital
and to commercialize the Products may be materially impacted by the COVID-19 pandemic.
The Company’s ability to raise capital and
to commercialize the Products may be materially impacted by the COVID-19 pandemic. The full impact on the economy and the capital
markets in the U.S. and the rest of the world from the COVID-19 pandemic are uncertain, in terms of both scale and duration. The
high level of volatility in the capital markets may make it difficult to raise funds, especially for early stage companies that
involve higher risk. If the Company raises sufficient funds to begin the work of commercializing Company products, the Company may
have difficulty securing supplies needed or manufacturing and distribution partners. The impact of social distancing measures and
related workforce reductions may negatively impact the ability of suppliers to deliver the Company to the components the Company
needs for manufacture or the ability of any of the Company’s potential partners to operate effectively to meet Company
requirements. In addition, many of the third parties that the Company would rely on for production and distribution are likely to be
highly engaged in manufacturing products aimed at combatting the pandemic by manufacturing testing supplies and equipment, medical
equipment and/or potential treatments. The Company cannot assure an investor that, should they raise sufficient funds, they will be
able to contract with suppliers, manufacturing partners or distribution partners at a level that would allow the Company to achieve
profitability, or at all.
The Company’s products may not achieve
market acceptance thereby reducing the chance for success.
The Company is only in the early stages of selling
its Products. It is unclear whether these Products or other unanticipated events may result in lower sales than anticipated, which could
force the Company to limit expenditures on research and development, advertising, and general Company requirements for improving and expanding
product offerings. The Company cannot guarantee consumer demand or interest in current or future product offerings, which could have a
material adverse effect on business, results of operations, and overall financial condition.
If the market chooses to buy competing Products, the Company may
fail.
Although the Company believes that its Products will
be commercially viable, there is no verification by the marketplace that the Products will be accepted by or purchased by customers at
the scale desired by the Company. Even though competing drugs for Central Nervous System (“CNS”) diseases are few, the market
may choose these competing drugs. If the market chooses to continue to use competing drugs, it may be more difficult for the Company to
ever become profitable which would be substantially harm the business and, possibly, cause it to fail whereby the Investors could lose
their entire investment. In addition to Company dependency on their continued services, Company future success will also depend on the
ability to attract and retain additional future key personnel. The Company may face intense competition for such qualified individuals
from well-established and better financed competitors. The Company may not be able to attract qualified new employees or retain existing
employees, which may have a material adverse effect on the Company’s results of operations and financial condition.
The pharmaceutical and medical device industries are subject to rapidly
changing technology which could make the Products and other products the Company develops obsolete.
The Company’s industry is characterized by
rapid technological changes, frequent new product introductions and enhancements and evolving industry standards, all of which could
make the Company’s Products obsolete. The Company’s future success will depend on its ability to anticipate and keep
pace with the evolving needs of the industry on a timely and cost-effective basis and to pursue new market opportunities that
develop as a result of technological and scientific advances. The attractiveness of the Company’s Products partly depends on
the ability to continue to improve the Products. Failure to deliver such improvements in the timelines suggested may affect the
Company’s business plan and ability to obtain greater market penetration, or otherwise cause the Company to lose market
share.
The Company will require additional funding
to develop and commercialize the Products. If the Company is unable to secure additional financing on acceptable terms, or at all, the
Company may be forced to modify its current business plan or to curtail Company planned operations.
The Company anticipates incurring significant operating
losses and using significant funds for the execution of the clinical trials, marketing the Products, and manufacturing capability scaling.
The Company’s existing cash resources are insufficient to finance these operations. Accordingly, the Company will need to secure
additional sources of capital to develop its business and the Products as planned. In the event that the Proceeds from this Offering are
not sufficient to successfully commercialize the Products, the Company may seek to secure future capital. The Company may seek substantial
additional financing through public and/or private financing, which may include equity and/or debt financings, and through other arrangements,
including collaborative arrangements. As part of such efforts, the Company may seek loans from certain of their executive officers, directors
and/or current shareholders.
If the Company is unable to secure additional financing
in the near term, the Company may be forced to: (1) Curtail or abandon the Company’s existing business plans; (2) Default on any
debt obligations; (3) File for bankruptcy; (4) Seek to sell some or all of Company assets; and/or (4) Cease business operations.
If the Company is forced to take any of these steps,
the Company’s common stock may lose significant value or become worthless.
Any future financing may result in ownership
dilution to the Company’s existing Shareholders and may grant rights to future investors more favorable than the rights currently
held by the Company’s existing Shareholders.
If the Company raises additional capital by issuing
equity, equity-related, or convertible securities, the economic, voting and other rights of the Company’s existing shareholders
may be diluted, and those newly-issued securities may be issued at prices that are at a significant discount to current and/or then fair
market value of the Shares. In addition, any such newly issued securities may have rights superior to those of the Company’s Common
Stock as offered through this Offering. If the Company obtains additional capital through collaborative arrangements, the Company may
be required to relinquish rights to technologies or product candidates than the Company might otherwise have or become subject to restrictive
covenants that may affect business.
Risks Related to Development and Regulatory Approval of The Company’s
Product
Defects or errors in the clinical trial stage
could set back the success of candidates, requiring further investment without a guaranteed outcome.
Even though the Company has been engaged in Phase
II and Phase IIB human clinical trials, the Company’s Products may contain undetected defects or errors when placed into further
human clinical trials, which could materially and adversely affect the Company’s reputation, result in significant costs to the
Company and impair their ability to sell their products in the future. The costs incurred in correcting any defects or errors may be substantial
and could adversely affect operating results. Furthermore, the Products may contain undetected defects or errors when first introduced
to the market or as new versions are released, which could materially and adversely affect the Company’s reputation, result in significant
costs to the Company and impair their ability to sell Company products in the future. The costs incurred in correcting any defects or
errors may be substantial and could adversely affect operating results.
The FDA, other regulatory bodies and other comparable
foreign regulatory authorities each have substantial discretion in the approval process and may either refuse to consider the company
application for review or may form the opinion after review of the data that the Company’s application is insufficient to grant
approval for commercial use.
Commercialization of the Products will require FDA
and other regulatory approval in the future. The FDA and other comparable foreign regulatory authorities each have substantial discretion
in the approval process and may either refuse to consider the business’s application for human clinical trials or may form the opinion
after review of the Company’s clinical trial data that the application for the Intrulin (and perhaps the CPD technology) is insufficient
to allow approval of for human health applications. Approval procedures vary among jurisdictions and can involve requirements and administrative
review periods different from, and greater than, those in United States. Moreover, any approvals that the Company obtains may not cover
all of the clinical indications for which the Company will seek approval, or could contain significant limitations in the form of narrow
indications, warnings, precautions or contraindications with respect to conditions of use. In such an event, the Company’s ability
to generate revenues from the Products would be greatly reduced and its business would be harmed.
Failure to obtain approval to market the Company
product for human health applications may limit Company prospects for growth.
The Company will require approval from the FDA and
similar agencies in other countries prior to marketing Company Products for human use. The Company will need to establish, to the satisfaction
of those organizations, that their products are safe and effective for use. Because the Company’s Products are novel and represents
a departure from the status quo, the Company cannot assure Investors that the Company will receive approval. In addition, the Company
believes that the resources of the FDA are heavily engaged in monitoring, reviewing and approving testing solutions for COVID-19 and the
underlying virus, SARS-CoV-2, and they may not have sufficient staffing or other resources to review the Company’s application in
a timely manner. As a result, the Company may not be in a position to pursue human health applications for a significant period of time.
The regulatory approval process of the FDA
for the Products and other comparable domestic and foreign regulatory authorities are lengthy, time-consuming and inherently
unpredictable, and if the Company is unable to obtain timely regulatory approval for the Products, the business will be
substantially harmed.
The Company is not permitted to market the Products
in the US until approval from the FDA, or in any foreign countries until the Company receives the requisite approval from such countries.
Prior to submitting for FDA for approval of the Products for human health applications, the Company will need to initiate and complete
at least one Phase III clinical trial for the Intrulin – and, potentially, clinical trials for the CPD technology. Successfully
completing the Company’s clinical program and obtaining approval is a complex, lengthy, expensive and uncertain process, and the
FDA may delay, limit or deny approval of the Company’s Products for many reasons, including, among others, because:
(1) The Company may not be able to demonstrate
that their product candidates are safe and effective in treating patients to the satisfaction of the FDA or other applicable foreign regulatory
agencies;
(2) The results of the Company’s
clinical trials may not meet the level of statistical or clinical significance required by the FDA or other applicable foreign regulatory
agencies for marketing approval;
(3) The FDA or other applicable foreign
regulatory agencies may disagree with the number, design, size, conduct or implementation of the Company’s clinical trials;
(4) The FDA or other applicable foreign
regulatory agencies may require that the Company conduct additional clinical trials;
(5) The FDA or other applicable foreign
regulatory authorities may not approve the labeling or specifications of the Company’s product candidates;
(6) The contract research organizations
(CROs) and other contractors that the Company may retain to conduct clinical trials may take actions outside of Company control that materially
adversely impact the clinical trials;
(7) The FDA or other applicable foreign
regulatory agencies may find the data from preclinical studies and clinical trials insufficient to demonstrate that the Products are safe
and effective for its proposed indications;
(8) The FDA or other applicable foreign
regulatory agencies may disagree with the Company’s interpretation of data from their preclinical studies and clinical trials;
(10) If and when the applications for regulatory
approval are submitted to the FDA or other foreign regulatory agencies, as applicable, the regulatory authorities may have difficulties
scheduling the necessary review meetings in a timely manner, may recommend against approval of the Company’s application or may
recommend or require, as a condition of approval, additional preclinical studies or clinical trials, limitations on approved labeling
or distribution and use restrictions;
(11) The FDA or other applicable foreign
regulatory agencies may change their approval policies or adopt new regulations.
Any of these factors, many of which are
beyond Company control, could increase development costs, jeopardize the Company’s ability to obtain regulatory approval for and
successfully market the Products and generate revenue. Moreover, because their business is entirely dependent on the approval of the
Products by the FDA and other foreign agencies, any such setback in the Company’s pursuit of regulatory approval would have a material
adverse effect on business and prospects.
There may be a high rate of failure for the Products proceeding through
clinical trials.
There may be a high rate of failure for Products as candidates proceeding
through clinical trials. The Company may suffer significant setbacks in their clinical trials, similar to the experience of a number of
other companies in medical device industry, even after receiving promising results in earlier trials. Further, even if the Company views
the results of a clinical trial to be positive, the FDA or other regulatory authorities may disagree with their interpretation of the
data.
Clinical trials are expensive, time consuming, uncertain and susceptible
to change, delay or termination.
Clinical trials are expensive, time consuming and difficult to design and
implement. The result of a clinical trial may be undesirable and can result in a clinical trial cancellation or the need for re-evaluation
and supplementation. Even if the results of the clinical trials are favorable, the clinical trials are expected to continue for several
years and may even take significantly longer to complete. In addition, the Company, the FDA the various IRBs, or other regulatory authorities,
may suspend, delay or terminate the Company’s clinical trials at any time, for various reasons.
Risks Related to the Securities and the Offering
There is no current market for the Shares.
There is no formal marketplace for the resale
of Company Shares and none is expected to arise for the foreseeable future. Investors should assume that they may not be able to liquidate
their investment.
The Offering price of the Company’s
Shares was not established on an independent basis; the actual value of an investment may be substantially less than what Investor pays
for the securities.
The Company’s Board of Directors
established the Offering price of the Company’s Shares on an arbitrary basis. The selling price of the Shares bears no
relationship to the book or asset values or to any other established criteria for valuing Shares. Because the Offering price is not
based upon any independent valuation, the Offering price may not be indicative of the proceeds that an Investor would receive upon
liquidation. Further, the Offering price may be significantly more than the price at which the Shares would trade if they were to be
listed on an exchange or actively traded by broker-dealers.
Kurve has existing Patents that the Company
might not be able to protect.
The Company's most valuable asset is its intellectual
property. Kurve holds 17 United States utility patents with two currently pending. It is possible that competitors may attempt to misappropriate
or violate intellectual property rights owned by the Company. The Company intends to protect its intellectual property portfolio from
such violations, within the constraints of available resources. It is important to note that unforeseeable costs associated with such
practices may consume a significant portion of capital, which could negatively affect the Company’s research and development efforts
and business, in general. However, the Company has anticipated the possible need to protect its Patents from infringement and therefore
has allocated Proceeds from this Offering, in the form of Working Capital, to account for this potential event.
The Company is controlled by Marc Giroux,
Chairman/Board of Directors and Chief Executive Officer
The Company’s Chairman of the Board and
Chief Executive Officer is the beneficial owner of a majority of the Company’s Shares of Common stock as of the date of this Offering
Circular. Upon a successful Offering (where the Maximum Offering Amount is raised) Marc Giroux will still own a majority of the Company’s
voting stock. Investors in this Offering will not have a majority of voting Shares and therefore will not have the have the ability to
control a vote of the Shareholders without consensus from Marc Giroux. Marc Giroux, therefore, has complete control as to the direction
of the Company. There is a disproportionate reliance on Marc Giroux for the operation of the Company, and therefore a risk that the direction
of the Company may change if Marc Giroux is unable to perform his duties as Director and Officer.
Bonus Shares have a potentially dilutive effect.
As part of this Offering, the Company is offering
Bonus Shares to certain investors (See “Plan of Distribution” below). There is the potential that the Company sells the Shares
solely through investments subject to the award of Bonus Shares. In the event the Company sells the Shares solely through investments
subject to the award of Bonus Shares, the Company could only raise Proceeds equaling $20,000,000 or 50% of the Maximum Offering Amount.
In the event the Company sells the Shares in amounts less than the total number of Shares offered, the Bonus Shares may still have a dilutive
effect to where the Maximum Offering Amount is not reached.
The Company has outstanding convertible notes
held by Tom McDowell and Marc Giroux.
The Company has four convertible notes outstanding
to Marc Giroux and Tom McDowell, both Officers and Directors of the Company. The total debt owed on these convertible notes is $300,000.
The conversion price is set at $2.08. This means that Marc Giroux and Tom McDowell will, if the notes are converted, receive Shares at
a price below the price offered in this Offering. Alternatively, Marc Giroux and Tom McDowell could cause the Company to repay these convertible
notes using the Company’s cash.
DILUTION
On March 25, 2022 the Company executed the
Merger in which Kurve Technology, Inc. assigned its assets to Savile Therapeutics, Inc. which was in-turn acquired by the Company.
Prior to the Merger, the persons owning Founders’ Shares (Marc Giroux, Tom McDowell, Glenn Cornett included) agreed to keep
their proportionate ownership in the Company as they had owned in Kurve Technology, Inc. prior to the Merger. After the Company
increased the number of authorized Shares, the above-named persons received their proportionate number of Shares to maintain their
respective equity ownership percentages in the Company. The following table describes the number of Shares assigned to each
Officer:
| Name | |
Number of Shares |
| Marc Giroux | |
| 43,416,595 | |
| Tom McDowell | |
| 3,011,382 | |
| Glenn Cornett | |
| 2,506,975 | |
The above-named persons received these Shares pursuant
to their roles as founders in Kurve Technology, Inc. prior to May 2021 (one year before the date of this Offering Circular).
For a more detailed discussion please see “Legal
Proceedings and Merger” below.
PLAN OF DISTRIBUTION AND SELLING SECURITYHOLDERS
The Offering will be made through general solicitation,
direct solicitation, and marketing efforts whereby Investors will be directed to the Portal kurvetx.com/invest to invest. The Company
has engaged Rialto Markets, an independent FINRA broker-dealer to assist with the Share sales in exchange for a 3% commission fee on the
aggregate sales. The Offering is conducted on a best-efforts basis. No Commissions or any other renumeration for the Share sales will
be provided to the Company, the Directors, any Officer, or any employee of the Company, relying on the safe harbor from broker-dealer
registration set forth in Rule 3a4-1 under the Securities Exchange Act of 1934, as amended.
The Company will not limit
or restrict the sale of the Shares during this 12-month Offering. No market exists for the Shares and no market is anticipated or intended
to exist in the near future, therefore there is no plan to stabilize the market for any of the securities to be offered.
Directors, Officers, and employees of the
Company are primarily engaged in the Company’s business of developing and manufacturing medical devices, and none of them are,
or have ever been, brokers nor dealers of securities. The Directors, Officers, and employees will not be compensated in connection
with the sale of securities through this Offering. The Company believes that the Directors, Officers, and employees are associated
persons of the Company not deemed to be brokers under Exchange Act Rule 3a4-1 because: (1) no Director, Officer, or employee is
subject to a statutory disqualification, as that term is defined in section 3(a)(39) of the Exchange Act at the time of their
participation; (2) no Director, Officer, or employee will be compensated in connection with his participation by the payment of
commissions or by other remuneration based either directly or indirectly on transactions in connection with the sale of securities
through this Offering; (3) no Director, Officer, or employee is an associated person of a broker or dealer; (4) the Directors,
Officers, and employees primarily perform substantial duties for the Company other than the sale or promotion of securities; (5) no
Director, Officer, or employee has acted as a broker or dealer within the preceding twelve months of the date of this Offering
Circular; (6) no Director, Officer, or employee will participate in selling this Offering after more than twelve months from the
Effective Date of the Offering.
Rialto Markets LLC (“Rialto”) has agreed
to act as placement agent to assist in connection with this Offering. Rialto is not purchasing or selling any securities offered by this
Offering Circular, nor is it required to arrange the purchase or sale of any specific number or dollar amount of securities. However,
Rialto has agreed to use their best efforts to arrange for the sale of the Shares offered through this Offering Circular. In addition,
Rialto may engage other brokers to sell the securities on their behalf. Rialto will receive compensation for sales of the Shares offered
and sold through pursuant to this Offering at a rate of 3% of the Gross Proceeds for a maximum of $1,500,000. In addition, the Company
may pay Rialto 8% of the Gross Proceeds from the sale of up to $10,000,000 in Common Stock resulting from the direct selling efforts of
Rialto not to exceed $1,600,000
The Company will also publicly market the Offering
using general solicitation through methods that include e-mails to potential Investors, the internet, social media, and any other means
of widespread communication.
This Offering Circular will be furnished to prospective
investors via download 24 hours per day, 7 days per week on the Company’s website at kurvetx.com/invest and via of the EDGAR filing
system.
The following table shows the total discounts and
commissions payable to Rialto in connection with this Offering by the Company:
In the event that Rialto’s targeted selling
efforts lead to sales of up to $20,000,000 in Shares of Common Stock, Rialto will be entitled to 8.0% of the Gross Proceeds from the sale
of such shares of Common Stock not to exceed $1,600,000.
| | |
Price Per Share | |
Total Offering |
| Public Offering Price | |
$ | 5.80 | | |
$ | 50,000,000 | |
| Placement Agent Commissions | |
$ | 0.174 | | |
$ | 1,500,000 | |
| Proceeds, Before Expenses | |
$ | 5.626 | | |
$ | 48,500,000 | |
Other Terms
Rialto has also agreed to perform the following services
in exchange for the compensation discussed above:
- Act as lead broker for the Offering, coordinating
efforts of parties involved and providing regulatory guidance;
- Manage the back-end process of the Offering
Platform technology, Investors use to invest in the Offering;
- Reviewing marketing materials if requested;
- Performing AML/KYC checks on all Investors,
and;
- Providing other financial advisory services
normal and customary for Regulation A offerings and coordinate with the Company’s registered transfer agent and legal representatives.
In addition to the commissions described above, the
Company will also pay $5,000 to Rialto for out-of-pocket accountable expenses paid prior to commencing the Offering. This fee will be
used for the purpose of coordinating filings with FINRA (Form 5110). In addition, the Company will pay Rialto $5,000 consulting fee
upon the issuance of the FINRA No Objection Letter and a $5,000 Blue Sky filing service fee for managing the filings required for Blue
Sky regulations. The Company will forward the fees required for state notice filing fees, estimated to be approximately $13,000.
Assuming the full amount of the offering is raised, and that Rialto's targeted selling efforts lead to sales of $10,000,000, the
Company estimates that the total fees and expenses of the Offering payable by the Company to Rialto will be approximately $1,200,000. Maximum
expected out of pocket expenses total $21,000.
Selling Securityholders
| Name of Selling Shareholder | |
Shares Owned Prior to Offering | |
Shares Offered Pursuant to this Offering | |
Shares Remaining after successful Offering |
| Marc Giroux(1) | |
| 43,416,595 | | |
| 397,778 | | |
| 43,018,817 | |
| Tom McDowell(1) | |
| 3,011,382 | | |
| 90,339 | | |
| 2,921,043 | |
| Glenn Cornett(1) | |
| 2,506,975 | | |
| 75,209 | | |
| 2,431,766 | |
| William Shankle(1) | |
| 1,254,742 | | |
| 37,642 | | |
| 1,217,100 | |
| David Dickstein(2) | |
| 40,000 | | |
| 20,000 | | |
| 20,000 | |
| Dan Bonfiglio(2) | |
| 20,000 | | |
| 10,000 | | |
| 10,000 | |
| Nilo Ramos(2) | |
| 20,000 | | |
| 10,000 | | |
| 10,000 | |
| Ted Esau(2) | |
| 12,019 | | |
| 6,009 | | |
| 6,010 | |
| Steve Salyer(2) | |
| 26,442 | | |
| 13,221 | | |
| 13,221 | |
| LSI(3) | |
| 1,000,000 | | |
| 250,000 | | |
| 750,000 | |
| Medical Funding Professionals(3) | |
| 900,000 | | |
| 225,000 | | |
| 675,000 | |
| Uhuru Network(3) | |
| 250,000 | | |
| 62,500 | | |
| 187,500 | |
The total number of Shares
offered by Selling Shareholders is 1,306,862. This represents 2.49% of all Shares outstanding as of the date of this Offering
Circular.
The Selling Shareholders are
entitled to sell their shares at different times in the Offering. The following is a synopsis of when the Selling Shareholders are entitled
to sell their Shares.
(1) Selling Shareholders are
entitled to sell their Shares once gross Proceeds reaches $25,000,000 or more.
(2) Selling Shareholders are
entitled to sell their Shares once gross Proceeds reaches $500,000 or more.
(3) Selling Shareholders are
entitled to sell the following proportion of Shares according to the following schedule:
Gross Proceeds of $10,000,000
– 6% of Shares* (as owned by that Selling Shareholder)
Gross Proceeds of $25,000,000
– 6% of Shares* (as owned by that Selling Shareholder)
Gross Proceeds of $35,000,000
– 6% of Shares* (as owned by that Selling Shareholder)
Gross Proceeds of $45,000,000
– 7% of Shares* (as owned by that Selling Shareholder)
*refers only to Shares offered
pursuant to this Offering
Bonus Program
Certain Investors will be eligible to receive
additional Shares of Common Stock (“Bonus Shares”) depending upon the amount invested by such Investors.
First 30 days of Offering –
Less than $100,000 investment
The Company will be offering 15% bonus in the
form of Bonus Shares for Investors that invest less than $100,000 within the first thirty (30) days following Qualification of the Offering
(not including the date on which the Offering is qualified).
First 30 days of Offering –
More than $100,000 investment
The Company will be offering 30% bonus in the
form of Bonus Shares for Investors that invest more than $100,000 within the first thirty (30) days following Qualification of the Offering
(not including the date on which the Offering is qualified).
After first 30 days of Offering –
More than $100,000 investment
The Company will be offering 20% bonus in the
form of Bonus Shares for Investors that invest more than $100,000 after the first thirty (30) days following Qualification of the Offering
(not including the date on which the Offering is qualified).
The Company will absorb the
cost of the issuance of the Bonus Shares; to the extent any are issued, it will reduce the Proceeds that the Company receives. The issuance
of these Bonus Shares will have a maximum potential dilutive effect of 28,735,632 Shares.
USE OF PROCEEDS
The Company intends to use the Proceeds of this Offering
to (1) Complete the purchase of select intellectual property from Kurve Technology, Inc. (through Savile Therapeutics, Inc.).; (2) Design,
develop, and execute human clinical trials for the Company’s Products; (3) Develop relationships with select producers of CNS drugs
for potential licensing relationships; (4) Expand research and development activities for the Company’s Products; (5) Gain FDA Approval/authorization
for the Company’s Products; (6) Marketing the Offering and the Company’s Products; (7) Selling Shareholders.
| | |
| 40% | | |
| 60% | | |
| 80% | | |
| 100% | |
| 1. Asset Purchase | |
| $4,600,000 (23%) | | |
| $4,600,000 (15.33%) | | |
| $4,600,000 (11.5%) | | |
| $4,600,000 (9.2%) | |
| 2. Marketing | |
| $3,300,000 (16.5%) | | |
| $4,800,000 (16%) | | |
| $2,500,000 (6.25%) | | |
| $2,500,000 (5%) | |
| 3. Contract Manufacturing/Add’l Applications | |
| — | | |
| — | | |
| — | | |
| $3,000,000
(6%) | |
| 4. Clinical Studies | |
| $6,000,000 (30%) | | |
| $6,000,000 (20%) | | |
| $20,000,000 (50%) | | |
| $25,000,000 (50%) | |
| 5. Working/Reserve Capital | |
| $5,000,000 (25%) | | |
| $8,600,000 (28.67%) | | |
| $6,200,000 (15.5%) | | |
| $7,300,000 (14.6%) | |
| Selling Shareholders | |
| $1,100,000 (5.5%) | | |
| $6,000,000 (20%) | | |
| $6,700,000 (16.75%) | | |
| $7,600,000 (15.2%) | |
| Total | |
| $20,000,000 | | |
| $30,000,000 | | |
| $40,000,000 | | |
| $50,000,000 | |
1. Asset Purchase
The Company is currently the debtor on a promissory
note with a principal amount of $4,600,000. This promissory note was made by Savile Therapeutics, Inc. and assumed by the Company pursuant
to the Merger (see “Description of the Business” below) for the purchase of all relevant assets from the Company’s predecessor
Kurve Technology, Inc. The Company will use the Proceeds of this Offering to pay off this promissory note. For a more detailed discussion
please see “Legal Proceedings and Merger” below.
2. Marketing
The Company intends to spend $7,450,000 on the costs
of (1) marketing the Shares in this Offering to Investors; and (2) marketing for the Company’s Products. The Company intends to
market its Products through the use of independent representatives and direct marketing to medical professionals and select pharmaceutical
companies.
3. Manufacturing
The Company intends to spend approximately
$1,000,000 on contract manufacturing of the Company’s Products concurrent with the Phase III trial (see “Description of
the Business” below). As the Phase III trial concludes and the Company approaches commercialization of the Products, it is
likely that reserve capital will be utilized to scale up production of the Products. Contract manufacturing will be an advantageous
path for state of the art precision robotic manufacturing utilizing the most current manufacturing technology to ensure full
compliance with Pharmaceutical and Medical Device Good Manufacturing Practices as required by the FDA.
4. Clinical Studies
The Company plans to use a portion of the Proceeds
to engage the FDA and conduct Phase II and Phase III human use clinical trials for its Products in several key Central Nervous System
(“CNS”) diseases. Depending on the level of funding secured, these include three studies that would launch under a Kurve Therapeutics
label.
| 1. | Alzheimer’s Disease (pAb) Phase II = $5M |
| 2. | Alzheimer’s Disease (insulin) Phase III = $20M |
The Company’s products are already participating
in 15 ongoing clinical trials not administered or run by the company. The Company also intends on participating in the clinical trials
of its pharmaceutical partners as the study device supplier as well as the high-volume manufacturer at commercialization.
5. Working/Reserve Capital
The Company intends to use $11,950,000 of the Proceeds
for Working Capital and Reserve Capital. Included in this use is payment of salaries, benefits, legal and other professional services,
and other administrative costs. As the Phase III trial concludes and the Company approaches commercialization, it is likely that reserve
capital will be utilized to scale-up production of the Company’s Products.
DESCRIPTION OF THE BUSINESS
Corporate History
Kurve Therapeutics, Inc. was founded as a Delaware
corporation on November 20, 2019. On January 25, 2022, the Company submitted an Amended and Restated Certificate of Incorporation, as
corrected, authorizing the Company to issue 75,000,000 Shares – 74,000,000 Common Shares and 1,000,000 Preferred Shares. As of the
Date of this Offering Circular, no Preferred Shares are issued or outstanding. The Company is offering 8,620,689 Common Shares through
this Offering.
On March 25, 2022, the Company merged with Savile
Therapeutics, Inc. Savile Therapeutics, Inc. was a non-operating holding company created for the purpose of holding assets once owned
by Kurve Technology, Inc. For a more detailed discussion please see “Legal Proceedings and Merger” below. The Agreement and
Plan of Merger between Savile Therapeutics, Inc and Kurve Therapeutics, Inc. has been included in and attached hereto as Exhibit 16B.
Prior to this Offering, the Company entered into a certain contract for
services with Medical Funding Professionals, MFP LLC DBA MFP, and Life Science Intelligence, Inc. (“LSI”) whereby MFP and
LSI provide advice and assistance in conjunction with the analysis of the Company’s economics and governance capital planning. These
advisors intend on selling a portion of their Shares through this Offering. See “Selling Shareholders” above.
Summary of the Company and Its Products
The Company has two main products. The first product
is Intrulin (Kurve intranasal insulin), which is a drug the Company is developing to combat cognitive aging, neurodegenerative diseases
and related disorders. The second product is the Company’s patented Controlled Particle Dispersion® (“CPD”) technology.
The retail name of the device utilizing the CPD technology is ViaNase®.
The Company intends to use the Proceeds of this Offering to (1) Complete
the purchase of select intellectual property from Kurve Technology, Inc.; (2) design, develop, and execute human clinical trials for the
Company’s Products; (3) Develop relationships with select producers of CNS drugs for potential licensing relationships; (4) Expand
research and development activities for the Company’s Products.
The Company and Its Products
Kurve Therapeutics is developing disease-modifying
drugs with demonstrated clinical efficacy in cognitive aging, neurodegenerative diseases and related disorders. Much of this efficacy
can be traced to inhibition of two key enzymes that have for over a decade been an elusive target for Alzheimer’s drugs: Glycogen
synthase kinase-3 beta (“GSK-3 beta”) and elongation factor 2 kinase (“EF2K”).
The CPD technology and the Intrulin (collectively
the “Technology” or “Products”) are intended to be used together - meaning the Intrulin’s delivery by ViaNase
increases the efficacy of GSK-3 beta and EF2K and avoids systemic side-effects by delivering drugs directly to the brain. This is done
with precisely-controlled, turbulent flow from an intranasal device.
The largest body of clinical evidence for the Technology
is for Alzheimer’s disease (a severe form of cognitive aging), where the Company has significant, placebo-controlled results in
five Phase II trials.
The Company has found that Intrulin increases blood
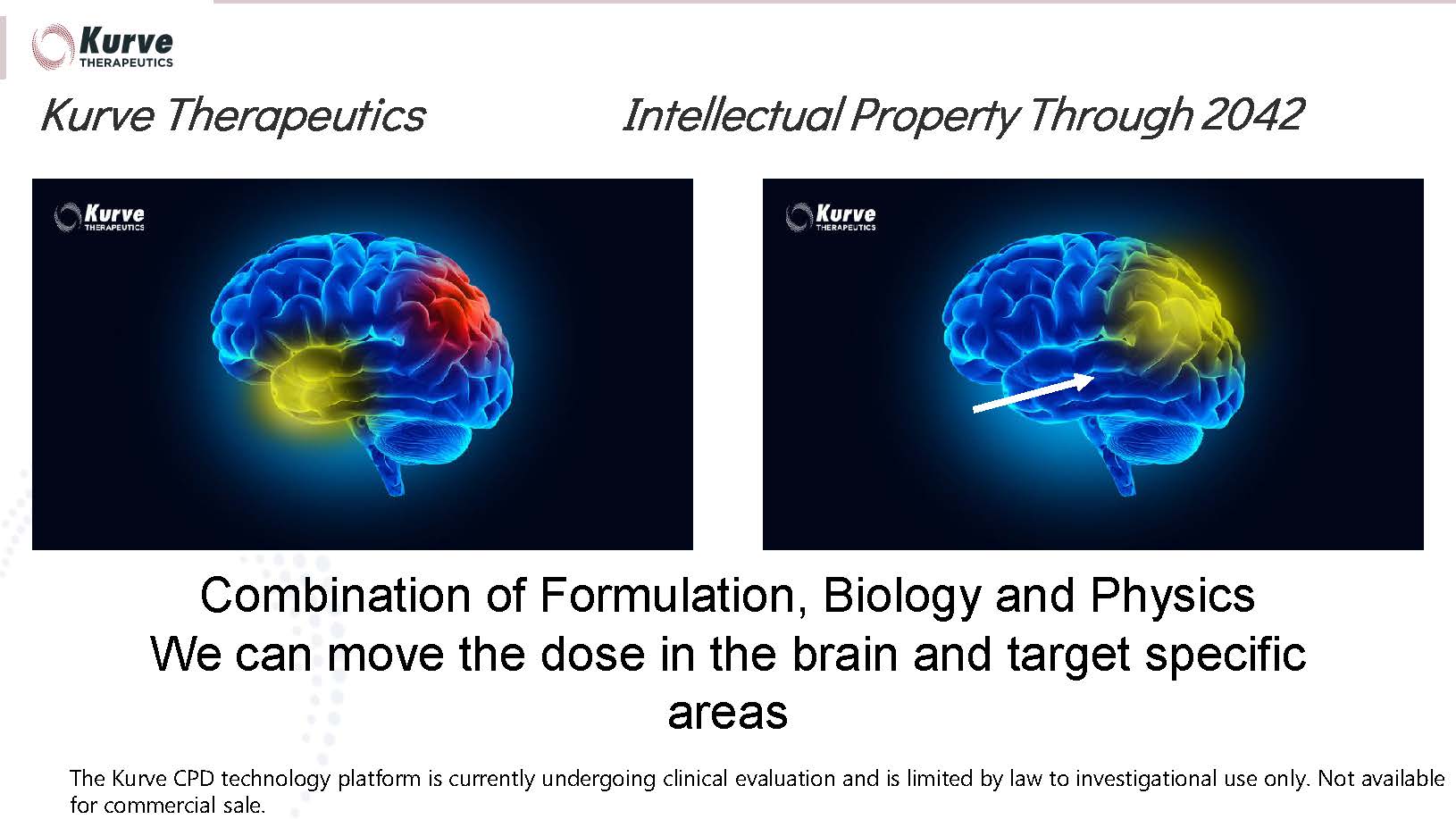
flow to cerebral gray matter in human patients. Kurve’s patented CPD technology enhances significantly the efficacy and safety
of existing and new formulations. This is done by accessing the most-penetrable part of the blood-brain barrier (“BBB”), the
olfactory mucosa at the top of the nasal cavity, where nerves penetrate to convey smell signals to the brain. This approach, known as
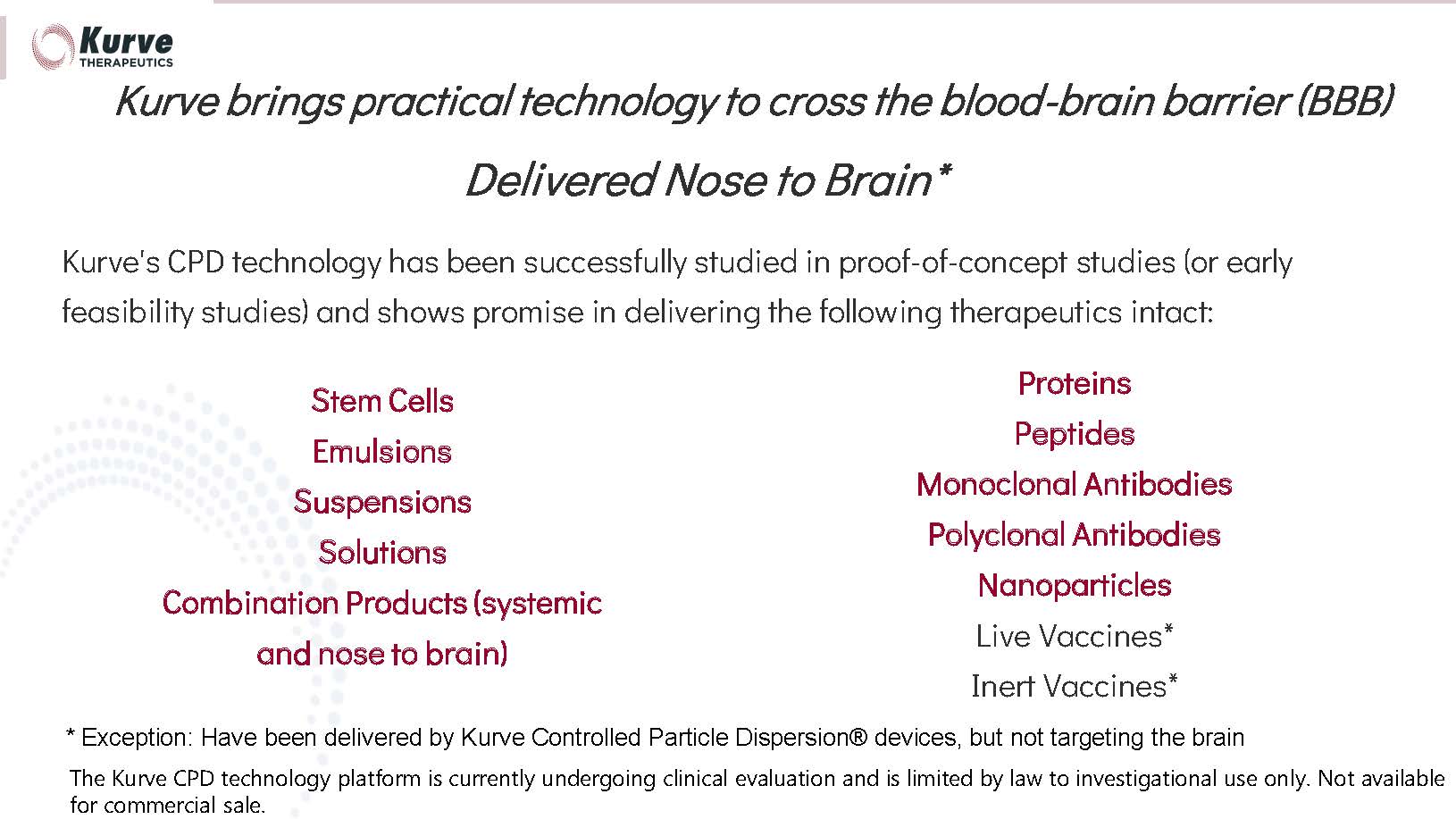
the nose-to-brain route, is particularly important for large, complex molecules (e.g., proteins, stem cells, and antibodies) that have
shown promise in neurodegenerative disease but are virtually impossible to deliver to the brain via other non-surgical routes (oral, intravenous,
etc.)
Kurve CPD’s precisely-controlled turbulent flow
generation, unlike other technologies, gets medicines past the curved surfaces inside the nose and up to the olfactory mucosa. This capability
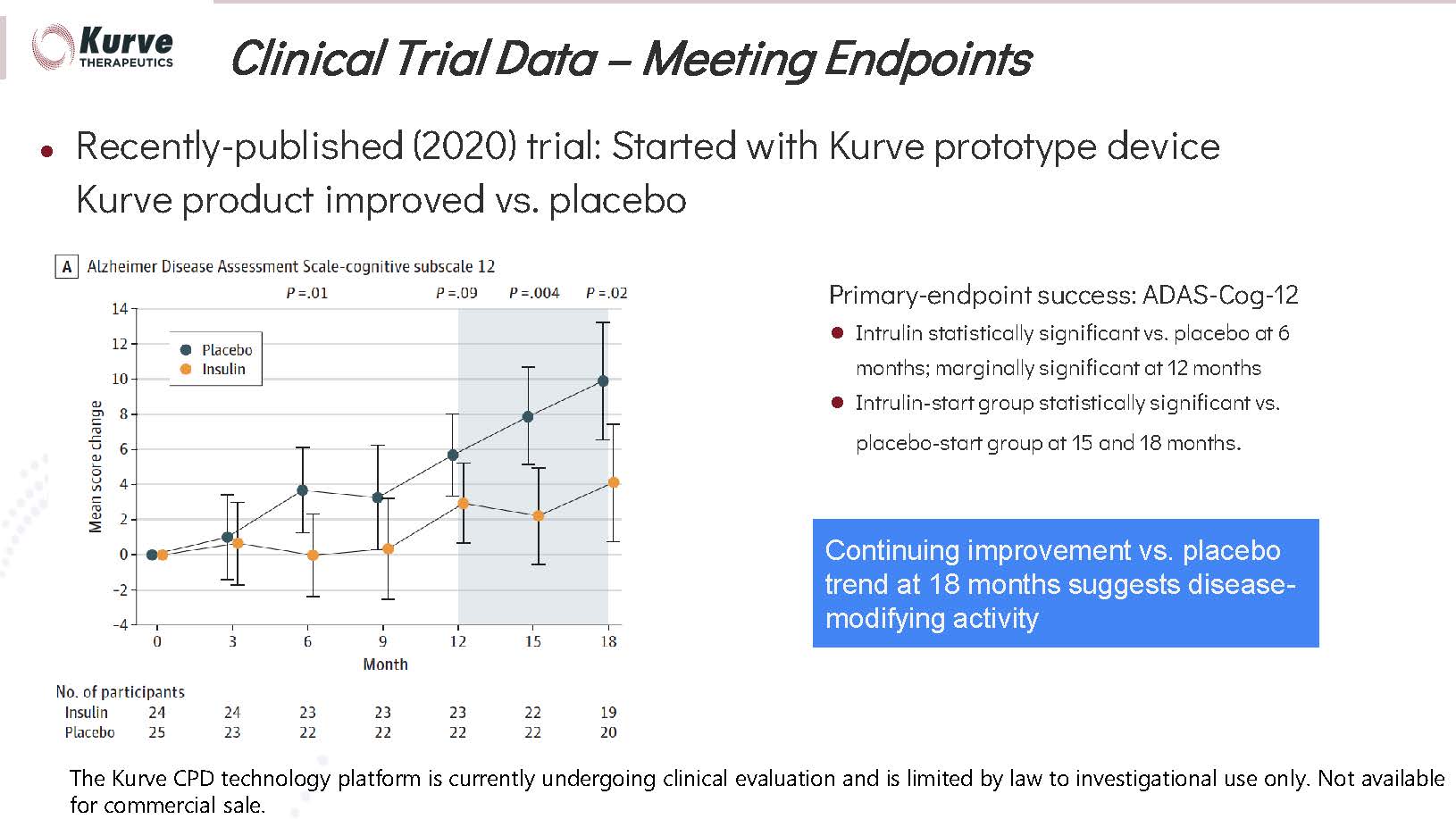
was demonstrated in an 18-month Phase II Alzheimer’s disease study using two different nasal delivery technologies. Patients using
the Kurve device showed improvement vs. baseline and placebo; the patients using the other device showed no improvement from placebo.
Kurve products have favorable clinical evidence of
disease-modifying activity in at least four neurodegenerative diseases. In a smaller, four-month Phase II study using positron
emission tomography ”PET” scanning, Alzheimer’s patients using the Technology demonstrated enhanced activity
vs. placebo in compromised brain regions. Marketed drug products are of limited efficacy, sometimes having benefits only briefly (3-6
months), after which patient decline accelerates and the use of the drug is discontinued. With Kurve, efficacy has continued for years
in individual patient cases and in a number of cases actually reversed the course of the disease. The longest clinical trial yet showed
continuing efficacy vs. the placebo group for 18 months. Kurve offers significant potential to advance the standard of care.
Market for the Company’s Products
The Company’s Products, Intrulin and ViaNase, are being developed
to combat cognitive aging, neurodegenerative diseases and related disorders. These disorders include (1) Alzheimer’s Disease; (2)
Parkinson’s Disease; (3) Lewy Body Dementia; and, (4) Amyotrophic Lateral Sclerosis (“ALS”).
1. Alzheimer’s Disease
Alzheimer’s Disease, with United States prevalence
estimated at 6 million, is the only top-10 cause of death in the United States with no known cure. It is the sixth-leading cause of death
and is the fifth-leading cause of death among those age 65 and older in the US. It is also estimated to be the most-expensive disease
in the nation. The 2018 cost of caring for Americans with Alzheimer’s and other dementias is about $277 billion, not including unpaid
caregiving by 16 million Americans annually providing an estimated 18.5 billion hours, valued in aggregate at $234 billion. Approved treatments
address only symptoms; they do not modify the disease in a fashion seen in some Kurve Therapeutics patient’s cases and clinical
trials. Global revenues for Alzheimer’s drugs were $18 billion in 2018. There is opportunity to grow the market significantly with
an effectively-delivered, disease-modifying drug.
2. Parkinson’s Disease
Parkinson’s Disease afflicts about 930,000 United
States patients, with 2030 prevalence forecast to grow to 1.2 million. While for some patients the symptoms can be treated effectively
for years, there are no approved drugs that treat the underlying neurodegenerative process. Global revenues for Parkinson’s drugs
were $4.5 billion in 2018. There is substantial opportunity to enlarge this market with a therapy yielding the disease-modifying experiences
seen in some Kurve patients.
3. Lewy Body Dementia
Lewy Body Dementia (“LBD”) affects about
1.4 million individuals and their families in the United States. It is the third-most-common dementia in the United States, after Alzheimer’s
and vascular dementia. There are no approved treatments demonstrated to slow or stop the brain cell damage caused by Lewy body dementia.
Global LBD drug revenues were $3.5 billion in 2018. Several LBD patients have benefitted significantly from Kurve treatments (patient
case reports pending).
4. Amyotrophic Lateral Sclerosis
Amyotrophic Lateral Sclerosis (“ALS”)
is a rare disease, with about 16,000 patients in the US. There are only two drugs approved for the treatment of ALS: Sanofi’s Rilutek
(riluzole) and Mitsubishi Tanabe’s Radicava (edaravone). The drugs were special cases in terms of FDA approval; they did not meet
the standard regulatory requirements for approval, but due to the increase in survival of patients, they were granted approval. Drug
sales for ALS in 2017 were collectively about $200 million in the US, Japan and major European markets (Germany, France, UK, Italy and
Spain).
Company Intellectual Property
Patents
The Company currently owns 17 issued utility patents
with the following Agencies (1) United States Patent and Trademark Office (“USPTO”) (2) Patent Cooperation Treaty; (3) European
Patent Office; (4) German Patent and Trademark Office; (5) French Patent and Trademark Office; (6) United Kingdom Intellectual Property
Office; and (7) Japan Patent Office. The Company currently has several patents pending as well. These patents represent the core technology
behind Intrulin and ViaNase. The earlier patents are in effect until 2027 with further patents in place until 2032; further patents are
pending.
Trademarks
The Company currently owns registered trademarks for
Kurve Therapeutics, Kurve Technology, ViaNase, and Intrulin.
Plan of Distribution of the Products
After approval by the relevant authorities, the Company
intends to initially distribute the Products in the United States and Europe. Within the United States, the Company will engage in direct
sales to customers. The customers in the United States primarily consist of hospitals, doctors, and hospital systems. Generally speaking,
the prescribing doctors have the discretion on whether to prescribe drugs. Once the decision has been made to prescribe the drug, the
doctor will direct the patient to purchase the drug via their pharmacy.
The Company intends to utilize distributors to
affect international sales of the drug. International sales methods of the Products will vary from country to country depending on
the respective laws and healthcare systems. International distributors and partners will be responsible for maintaining sales and
regulatory compliance within each international market. Additionally, the Company intends on using some of the Proceeds for
international marketing efforts.
Total Number of Employees
As of the Date of this offering Circular, the Company
has one (1) full time employee and no part time employees. However, the Company does utilize the services of independent contractors
for much of its operations. The Company intends on hiring one or more of these independent contractors if a sufficient amount of the Proceeds
are raised pursuant to this Offering.
Legal Proceedings and Merger
Kurve Therapeutics, Inc. (the “Company”)
is a successor entity to two former entities, Savile Therapeutics, Inc. and Kurve Technology, Inc.
Kurve Technology was the original operational company
which developed the Company’s Products. In January 2017, an investor in Kurve Technology, Inc. (who owned a significant portion)
of Kurve Technology died. The beneficiaries of this person’s estate intended to divest their portion of Kurve Technology, Inc. along
with several other investments (with other unrelated companies) held within the deceased investor’s estate. Management decided that
a State of Washington receivership proceeding was the best strategy for handling the redemption of the equity portion in question.
Management executed the receivership process for Kurve
Technology, Inc. to resolve the issue with the estate beneficiaries. The various processes as described within this sub-section will hereinafter
be collectively referred to as the “Receivership”. Marc Giroux, as majority shareholder in Kurve Technology, Inc. and Kurve
Theraputics, Inc. was named as a party in the Receivership as a procedural matter.
On February 22, 2022 the Receivership court accepted
the plan put in place between Kurve Technology, Inc. and the receiver Resource Transition Consultants, LLC (the “Receiver”).
The Court issued an “ORDER APPROVING GENERAL RECEIVER’S MOTION TO SELL ASSETS FREE AND CLEAR” (the “Order”)
whereby the court found that the assignment of the Assets described transaction to Savile Therapeutics, Inc. (as described below) was
“in the best interests of the estate”. The Order is attached hereto as Exhibit 16A.
On March 8, 2022 the assets from Kurve Technology,
Inc. were transferred, via the Receiver to Savile Therapeutics, Inc. (“Savile”). Savile was an affiliate of the Company, owned
in full by Tom McDowell. Savile had no operating history. The only function of Savile was to exist as a transitory entity into which the
assets from the receivership could be held. As part of this transaction, Savile issued a $4,600,000 promissory note with the receiver
as the holder.
On March 25, 2022 the Company, Kurve
Therapeutics, Inc. executed and filed a Certificate of Merger with the State of Delaware, in which Savile was merged with the
Company with the Company being the surviving corporation. Through this merger, the Company received all of Savile’s assets and
liabilities. The documents affecting these transactions are attached hereto as Exhibit 16B.
The following assets were transferred from Kurve Technology, Inc. to
Savile, to the Company:
- All patents approved, in review, applied for, abandoned, and expired
collectively in the United States, Canada European Union and select members of the European Union, and Japan.
- All Trademarks approved, in review, applied for, and expired in the United
States
- Website and URL kurvetech.com
- All customer contracts
- All rights to results of drug trial and studies including publication
of trials and studies.
- All rights to publications mentioning Kurve Technology, Inc.
- Laboratory
- Lab equipment
- Tooling
- Prototypes
- Molds
- Raw materials
- Devices
- Production equipment
- Completed devices
- Production designs
- Laboratory Fixtures
- Office Furniture
- Data equipment
- Laboratory leases
None of the Directors or Officers have been through a bankruptcy proceeding. None of the Directors or Officers have any criminal convictions.
None of the Directors or Officers are currently party to any civil suits as of the Date of this Offering Circular.
Special Characteristics of the Company’s
Operations and Competing Products/Procedures
The Company is subject to regulation of its Products
before, during, and after approval by the FDA Products for commercial distribution of the Products
The Company’s products have two separate approval/authorization
paths, one for the Intrulin, the other for the ViaNase/CPD technology. The Intrulin is regulated as a “Drug” with the FDA,
while the ViaNase is a Class I Medical Device, until such is paired with the Intrulin. When paired with the Intrulin, the ViaNase becomes
a Class II medical device as a drug/device combination. Below is a discussion of the dual regulatory paths for the Company’s Products.
FDA Approval for the Intranasal Insulin (Intrulin)
The Company has developed and is currently testing
its new drug, Intrulin. The Company has already undergone Phase I and Phase II human clinical trials for the Intrulin product.
The Company is going to seek authorization from the FDA to conduct Phase III human clinical trials to be funded using the Proceeds of
this Offering. After the Phase III human clinical trials are completed the Company intends to submit a New Drug Application (“NDA”).
The clinical phase of drug development focuses on
testing new drugs on human volunteers. Phase I tests may involve dozens of volunteer subjects and are performed to ascertain if the new
drug has side effects and how the human body metabolizes and excretes the drug. Phase II tests may involve hundreds of volunteer subjects
and are used to gather preliminary data on the efficacy of the new drug on a specific condition or disease. These studies may compare
the efficacy of the new drug against established drugs used to treat a condition or against placebos. After Phase II trials are completed,
a sponsor will typically coordinated with the FDA to plan its Phase III trials. Phase III trials may involve thousands of volunteer participants,
as the new drug is tested to evaluate its effectiveness and safety in many different population subsets.
During the clinical phase of drug development, the
FDA may halt testing of a new drug, temporarily or permanently, if there are concerns for the safety of trial testing volunteers. Institutional
Review Boards (“IRBs”) are also involved in reviewing the safety protocols for these trials, and may halt testing of a new
drug.
Once Phase III tests are concluded, a sponsor prepares
and submits a New Drug Application with the FDA. The FDA must approve the NDA before marketing of the new drug is permitted in the United
States. The NDA contains the results of all new drug testing, including preclinical and clinical testing, along with other information
about the new drug, including its chemical composition and manufacturing processes. The costs associated with preparation and submission
of an NDA are significant, and the approval process can take years.
After an NDA is submitted, the FDA has 60 days to
assess if the NDA will be accepted for filing based whether the NDA is sufficiently complete. Upon acceptance of the filing by the FDA,
a 180-day period review begins during which time the FDA conducts an in-depth review of the NDA’s contents to evaluate a sponsor’s
research on the new drug’s safety and effectiveness. After the 180-day period ends, the FDA must either issue an approval letter
for the NDA or a response letter to a sponsor addressing particular concerns. This 180-day period may be modified; the FDA and a sponsor
may come to an agreement to alter the review period timeline.
The FDA subsequently reviews the labeling of the new
drug to ensure relevant health information is present and inspects a sponsor’s manufacturing facility where a new drug is produced
to confirm that the manufacturing of the new drug is compliant to standards. (See “Facilities Regulation” below)
After a sponsor’s new drug passes through these
steps, the new drug approval process is complete. A sponsor is required to provide the FDA with periodic updates regarding the safety
of the new drug.
FDA Approval for the ViaNase Medical Device
The Company believes that the ViaNase and the CPD
technology will be regulated in the United States similarly as Class II medical devices by the FDA under the Federal Food, Drug and Cosmetic
Act. The FDA classifies medical devices into one of three classes based upon controls the FDA considers necessary to reasonably ensure
their safety and effectiveness. Class I devices are subject to general controls such as labeling, adherence to good manufacturing practices
and maintenance of product complaint records, but are usually exempt from premarket notification requirements. Class II devices are subject
to the same general controls and also are subject to special controls such as performance standards and may also require clinical testing
prior to approval. Class III devices are subject to the highest level of controls because they are life-sustaining or life-supporting
devices.
The CPD Technology-based
products, including ViaNase are anticipated to be regulated as Class II medical devices. There are two regulatory paths to authorizing
a medical device to be sold commercially. First is the 510(k) Premarket Notification process, the second is the De Novo Process. The Company
may elect to undergo the FDA’s Pre-Submission Process prior to the authorization processes.
Pre-Submission Process
The
Company intends to go through the Pre-Submission Process with the FDA prior to making the determination on whether to file as a
Pre-Market Notification 510(k) Class II or De Novo device.
The Pre-Submission process
is appropriate for situations such as the following:
-The device involves new
technology, a new intended use, or a new analyte and it will be helpful to familiarize the FDA with the novel features in advance of the
submission;
-Assistance is needed in
defining possible regulatory pathways;
-The studies involve complex
data and/or statistical approaches;
-The predicate or reference
method is unclear or uncertain; or
-The new device is a multiplex
device capable of simultaneously testing a large number of analytes.
A sponsor should submit a
Pre-Submission if they would like the FDA's thoughts on their studies or proposals prior to starting the studies. The
potential benefits of submitting a Pre-Submission are:
-To begin a dialogue with
the FDA and promote greater understanding;
-To reduce the cost of research
studies by focusing in on the important information needed for the FDA's approval (or clearance) and eliminating unnecessary or burdensome
studies, and
-To facilitate the review process for the future
marketing application since the FDA will already be familiar with the device.
Pre-Submissions and associated
meetings are strictly voluntary, and any comments or recommendations made in the review of protocols or during these meetings are not
binding on the manufacturer or the FDA.
510(k) Premarket Notification
If, after the
Pre-Submission process, the FDA and Company deem that the device requires the submission of a Premarket Notification 510(k), the Company
will be prohibited from commercially distributing the device until the Company receives a letter of substantial equivalence from FDA authorizing
the Company to sell commercially. A 510(k) must demonstrate that the device is substantially equivalent to one legally in commercial distribution
in the United States: (1) before May 28, 1976; or (2) to a device that has been determined by FDA to be substantially equivalent (a “predicate
device”). Even though the CPD technology is a proprietary and more effective delivery system than any other intra-nasal delivery
system on the market, the Company believes that the ViaNase can be authorized as substantially-equivalent to one or more predicate devices.
The De Novo Classification
If the Company determines,
after the Pre-Submission process, that the ViaNase and CPD technology is not substantially equivalent to a predicate device, the Company
will pursue a De Novo Classification.
The De Novo process provides
a pathway to classify novel medical devices for which general controls alone, or general and special controls, provide
reasonable assurance of safety and effectiveness for the intended use, but for which there is no legally marketed predicate device.
De Novo classification is a risk-based classification process.
Devices that are classified
into Class II through a De Novo Classification Request (De Novo request) may be marketed and used as predicates for future 510(k) premarket
notification submissions, if necessary. This means that it may be prudent to initially pursue a De Novo Classification for the CPD technology
at this early stage, so the Company can file 510(k) premarket notifications for its future products based on that technology.
The De Novo Classification Request includes the following information:
(1) Administrative Information, such as the device's
intended use, prescription use or over-the-counter use designated, etc.
(2) Device Description, which includes but is
not limited to technology, proposed conditions of use, accessories, and components.
(3) Classification Information and Supporting
Data:
-Clinical data (if applicable) that are relevant
to support reasonable assurance of the safety and effectiveness of the device.
-Non-clinical data including bench performance
testing.
-Information on the reprocessing and sterilization,
shelf life, biocompatibility, software, electrical safety and electromagnetic compatibility, animal study, literature (if applicable);
and
-A description of the probable benefits of the
device when compared to the probable or anticipated risks when the device is used as intended.
The Company will be required
to collect and submit this data prior to submission.
Facilities Regulation
Regulation of the Company
as a Drug Manufacturer
The Company intends to outsource
manufacturing of the Intrulin to third party drug manufacturers that are themselves FDA-regulated drug manufacturers.
Quality System Regulation
and Good Manufacturing Practices for Medical Devices
After approval of the CPD technology-based products
by the FDA, the Company will be subject to FDA Quality Systems Regulation (“QS Regulation”) and Good Manufacturing Practices”
(“GMPs”) Manufacturers must establish and follow quality systems to help ensure that
their medical device products consistently meet applicable requirements and specifications known as current good manufacturing practices.
Continued Compliance with these QS Regulations and GMPs is an ongoing duty of the Company. On February 23, 2022, the FDA proposed a regulation
which would incorporate the international standard specific for medical device quality management systems set by the International Organization
for Standardization (ISO), ISO 13485:2016 Medical devices – Quality management systems – Requirements for regulatory purposes.
The Company intends to use contract manufacturers. All of these contract manufacturers will be required by the Company to be ISO 13485:2016,
and thus compliant with FDA GMP.
The Company
does not anticipate that this proposed regulation will affect the operations of the Company, since the Company intends on complying with
ISO 13485:2016 prior to the proposed rule change in development of its facilities.
Plan of Operations
- Apply for and Submit the Intrulin for Phase III Human Clinical Testing with the FDA;
- Seek 510(k) or De Novo authorization from the FDA for the CPD technology and ViaNase;
- Conduct clinical trials for the CPD technology and ViaNase (only if required by FDA);
- Conduct Human Clinical Trials; (approx. $50,000-$75,000 per patient in the clinical trial)
- Implement GMP and other manufacturing/control standards for Company and its facilities.
The Company anticipates this Plan of Operations will
take approximately $15,000,000-$25,000,000 to execute. The Company is not reasonably able determine the costs of each step at this time
due to the conditionality of the human clinical trial process(es).
Competition
The Company believes that the only truly competing products (ones that
are cleared by the FDA and that are commercially available) are infusion products such as: (1) Privigen from CSL Behring LLC; (2) Aduhelm
(Aducanumab) from Biogen Inc.; (3) Gammagard from Takeda Pharmaceuticals U.S.A., Inc. All of these products are infusions approved to
treat various forms of dementia.
DESCRIPTION OF PROPERTY
As of the Date of this offering Circular, the Company
does not own real property or business personal property of material significance.
MANAGEMENT’S DISCUSSION AND ANALYSIS
OF THE FINANCIAL CONDITION OF THE ISSUER
Kurve Therapeutics, Inc. is a privately-held entity
organized in November 2019 in the State of Delaware and registered as a foreign corporation in the State of Washington. The following
includes a discussion of both Kurve Technology, Inc., predecessor to the Issuer, and Kurve Therapeutics Inc. On March 8, 2022 Kurve Therapeutics
purchased all of the significant assets and operations of Kurve Technology, Inc. See “Description of the Business” above for
a detailed discussion of the Merger.
Components of Results of Operations
The following discussion of the financial condition
and results of operations should be read in conjunction with the financial statements and the related notes included elsewhere in this
Offering Statement. The following discussion contains forward-looking statements that reflect the Company’s plans, estimates and
beliefs. The Company’s actual results could differ significantly from those discussed in the forward-looking statements. Unless
otherwise indicated, the latest results discussed below are as of December 31, 2021. *Please note that the Issuer’s financial statements
will be forthcoming on the various periodic filings, if this Form 1-A is qualified by the SEC.
Revenue/Net Loss from Operating Activities
Prior to the Merger, Kurve Therapeutics, Inc. is,
and has been, solely focused on Central Nervous System and Neurodegenerative Diseases. As of the date of this Offering Circular, the Company
has had Revenues but has never been profitable.
The Revenues for Kurve Therapeutics, Inc. were generated
through the sale of ViaNase to clinical trial partners.
In FY2021 the Company had a Net loss of $65,427.
In FY2020 the Company had a Net loss of $80,725. This represents a decrease in Net loss of $15,298 or 18.95%. The reason for the
decrease in Net loss was due to increased Revenue resulting from the sale of Products to clinical trial partners concurrent with a
small increase in ViaNase production costs.
Kurve Technology, Inc. is, and has been, solely focused
on the development of the Technology for the Drug Delivery market. As of the date of this Offering Circular, the Company has never been
profitable. In FY2021 Kurve Technology, Inc. had a Net loss of $385,046. In FY2020 Kurve Technology, Inc. had a Net loss of $416,673.
This represents a decrease in Net loss of $31,627 or 7.59%. The reason for this decrease is a significant increase in revenue from clinical
partners and the sales of devices to individual patients under the compassionate use program.*
*When a doctor prescribes the CPD technology devices
prior to authorization by the FDA under the 510(k) or De Novo Authorization.
Prior to delivery of the Products (ViaNase or Intrulin)
to customers, Kurve Technology receives full payment from clinical partners and sales of devices to individual patients under the compassionate
use program. As of December 31, 2021 a clinical partner paid in full for the devices needed for their trial, but the devices were not
shipped until February 2022. The amounts collected prior to shipping were classified as Deferred Revenue.
Operating Expenses
Kurve Therapeutics, Inc. classifies its Operating
Expenses as wages & benefits, sales & marketing, research & development, office expenses, bad debt expense, conferences, travel
& entertainment, professional fees, and computer expenses. In FY2021 Kurve Therapeutics, Inc. had Operating Expenses totaling $128,344.
In FY2020 Kurve Therapeutics, Inc. had Operating Expenses totaling of $119,540. This represents an increase in Operating Expenses of $8,804
or 7.36%. The reason for this increase is due to higher Salaries & Wages and Sales & Marketing costs, offset by reduced research
& development costs.
Kurve Technology, Inc. classifies its Operating Expenses
as wages & benefits, research & development, professional fees, rent expense, office expenses, patent amortization, computer expenses,
travel expenses, marketing expenses, and bad debt expense. In FY2021 Kurve Technology, Inc. had Operating Expenses totaling $406,771.
In FY2020 Kurve Technology, Inc. had Operating Expenses totaling of $486,246. This represents a decrease in Operating Expenses of $79,475
or 16.34%. The reason for this decrease is primarily reduced research & development costs and lower amortization expense of patents.
Research and Development Expense
Prior to the Merger, Kurve Therapeutics, Inc.’s
research and development efforts for FY2020 and FY2021 were focused on improved device design, device optimization, and increasing access
to the brain through the olfactory region and trigeminal nerve.
In FY2021 Kurve Therapeutics, Inc. spent $7,254 on
research and development expenses. In FY2020 Kurve Therapeutics, Inc. spent $60,098 on research
and development expenses. This represents a decrease of $52,844 or 87.93%. The reason for this decrease is reduced research and development
spending activity in favor of other expenses.
In FY2021 Kurve Technology, Inc. spent $118,971 on
research and development expenses. In FY2020 Kurve Technology, Inc. spent $141,618 on research
and development expenses. This represents a decrease of $22,647 or 15.99%. The reason for this decrease is reduced research and development
spending activity in favor of other expenses.
The Company anticipates that it will increase these expenses significantly
upon a successful Offering as the Company will begin Phase II and Phase III human clinical trials (see “Use of Proceeds” above).
Wages and benefits
In FY2021 Kurve Therapeutics, Inc. spent $87,158
on wages and benefit expenses. In FY2020 Kurve Therapeutics, Inc. spent $29,918 on wages and benefit expenses. This represents an increase
of $57,240 or 191.32%. The reason for this increase is that the CEO, the lone employee’s expenses were borne by Kurve Therapeutics
in 2021.
In FY2021 Kurve Technology, Inc. spent $144,317 on
wages and benefit expenses. In FY2020 Kurve Technology, Inc. spent $157,702 on wages and benefit expenses. This represents a decrease
of $13,385 or 8.49%. The reason for this decrease is transfer of the lone employee’s wages and expenses from Kurve Technology to
the Company in 2021.
Shareholders’ Equity
Kurve Therapeutics, Inc.
On January 1, 2020 50,189,694 Shares of Kurve
Therapeutics, Inc. Common Stock were outstanding, with a total Stockholders’ Equity of $80,725. These shares were issued as
Founders’ Shares. All of these shares were issued to Marc Giroux, Tom McDowell, Glenn Cornett, and William Shankle. Issuer had
to increase its number of authorized Shares before it could issue all of the Shares to Marc Giroux, Glen Cornett, and William
Shankle. Tom McDowell received his Shares pursuant to the Merger.
On December 31, 2020 50,284,117 Shares of Kurve
Therapeutics, Inc. Common Stock were outstanding, with a total Stockholders’ Equity of $49,275. This represents an increase of
94,423 Shares. The reason for this increase was due to the sale of shares to investors for proceeds of $130,000 at prices starting
at $1.25 per share, increasing to $2.08 per share.
On December 31, 2021 50,308,155 Shares of Kurve
Therapeutics, Inc. Common Stock were outstanding, with a total Stockholders’ Equity of $33,848. This represents an increase of
24,038 Shares. The reason for this increase was due to the sale of shares to investors at $2.08 per share for proceeds of $50,000.
Kurve Technology, Inc.
On December 31, 2019 through December 31, 2021 29,595,456
Shares of Kurve Technology, Inc. were outstanding, 500,000 of which were Preferred Shares.
On December 31, 2019 the total
Stockholders’ Deficit was $4,145,489.
On December 31, 2020 the total
Stockholders’ Deficit was $4,562,162. This represents an increase of $416,673 or 10%. The reason for this increase was due to
accrued interest payable on loans to Shareholders.
On December 31, 2021 the total
Stockholders’ Deficit was $4,947,208. This represents an increase of $385,046 or 8.44%. The reason for this increase was
primarily due to accrued interest payable on loans to Shareholders.
Liquidity and Capital Resources
As of December 31, 2021 Kurve Therapeutics, Inc.
had cash reserves of $373,804, Total Assets of $640,574. On December 31, 2020, Kurve Therapeutics, Inc. had Cash of $46,300, total assets
of $87,026. This represents an increase in $327,504 (or 707.35%) in cash and $553,548 (or 636.07%) in total assets.
As of December 31, 2021 Kurve Technology, Inc. had
cash reserves of $16,355, total assets of $95,980. On December 31, 2020, Kurve Technology, Inc. had cash of $32,662, total assets of $123,449.
This represents a decrease in $16.307 (or 49.92%) in cash and $27,469 (or 22.25%) in total assets.
The Company has financed its operations primarily
through the sale of Products to clinical partners, sales of Common Shares to investors and the sale of convertible debt to lenders.
The Company will have additional capital requirements
during fiscal year 2022 to conduct the human clinical testing and execute the other anticipated uses of the Proceeds (see “Use of
Proceeds” above). Therefore, the Company will attempt to raise additional capital through the sale of its securities in this Offering.
The Company’s Offering is for a maximum of 8,620,689 Shares of its Common Stock at price of $5.80 per share, with potential aggregate
gross Proceeds of $50,000,000.
There can be no assurance that the Company will be
successful in acquiring additional funding at levels sufficient to fund its future capital requirements. If the Company is unable to raise
additional capital in sufficient amounts or on terms acceptable to it, the Company may have to significantly reduce, delay, scale back
or discontinue the development of its technologies and patents or discontinue operations completely.
DIRECTORS AND EXECUTIVE OFFICERS
Directors
| Name | |
Position | |
Age | |
Term of Office | |
Approx. Hrs. per week |
| Marc Giroux | |
| Director | | |
| 58 | | |
| January 2020 - Present | | |
Full Time |
| Thomas McDowell | |
| Director | | |
| 64 | | |
| January 2022 - Present | | |
Full Time |
Executive Officers
| Name | |
Position | |
Age | |
Term of Office | |
Approx. Hrs. per week |
| Marc Giroux | |
Chief Executive Officer
(“CEO”) | |
| 58 | | |
| January 2020 – Present | | |
Full Time |
| Thomas McDowell | |
Chief Financial Officer
(“CFO”) | |
| 64 | | |
| January 2022 – Present | | |
Full Time |
| Dr. Glenn Cornett, M.D., Ph.D. | |
Chief Science Officer
(“CSO”) | |
| 60 | | |
| January 2022 – Present | | |
Full Time |
Marc Giroux - Director and CEO
Education and Experience
Marc was most recently the President of EES, Inc.
– a high tech staffing company placing executives across the USA. EES’ main focus was consulting and building executive teams
from early stage through exit strategy. Marc is considered an industry expert on the formation and building of teams. Earlier in his career,
Marc spent 13 years in high-tech manufacturing where he developed and invented new products and designed efficiency equipment for production
manufacturing. A long-time rhinosinusitis and allergy sufferer, Marc invented the proprietary Kurve technology. He leads Kurve’s
business development efforts with investors and partners and guides the engineering team.
Duties of Marc Giroux as Director and CEO
- Establishing the Company vision
- Guiding the Company through the launch of the Products
- Managing the licensing, both in and out, with regard to drugs and devices
for therapies in neurodegenerative diseases and oncology
- Establishing partnerships with pharmaceutical companies
- Revenue and investment generation
Thomas McDowell, Director and CFO
Education and Experience
Tom holds a Bachelor of Science in Business Administration
and Accounting from the University of Southern California. He is an inactive CPA in Washington and California. He started his career in
public accounting and soon jumped into the world of working with entrepreneurial companies, helping them develop processes and ultimately
formulating exit strategies. That led to almost 15 years in investment banking and then about 10 years consulting with companies to help
stabilize their finance departments and then restructure and build into more forward looking groups that were not solely riveted on the
rear view mirror.
Duties of Thomas McDowell as Director and CFO
As a Director Tom is responsible for ensuring that
business operations are performed according to the duties and responsibilities set in the Bylaws. He will ensure that the Company complies
with all regulatory reporting. He will help to identify key team members to fill the roles that will build sales, develop formulations
to treat specific ailments, and create new technologies that will continue keep the Company at the forefront of nose to brain treatments
and drug delivery.
As the CFO, Tom will oversee all financial operations, compliance, and
to protect the integrity of all company assets.
Dr. Glenn Cornett, M.D., Ph.D., Chief Science Officer
Education and Experience
Dr. Glenn Cornett is a biotech entrepreneur and advisor.
He holds an M.D. with Distinction in Research from the University of Michigan and a Ph.D. in neuroscience from UCLA. He has spent the
past decade developing drugs for psychiatric and neurological indications.
Dr. Cornett’s experience includes:
-Eli Lilly: He started a research strategy unit and
developed a global financial model that was used for the Cialis acquisition.
-McKinsey: Dr. Cornett consulted to clients in high-tech,
heavy industry and health care sectors. He was in the system dynamics practice and a founding member of the complexity practice.
-Razorfish: He was a Vice President successively running
the US and European strategy practices, establishing new product-service offerings and developing new clients.
-Los Alamos National Laboratory: Dr. Cornett authored
a monograph on plutonium and public health policy while also working on further (confidential) efforts.
Dr. Cornett’s entrepreneurial spirit, passion
for innovative science and desire for humanitarian impact all contribute to his enthusiasm for Kurve Therapeutics.
Duties of Glenn Cornett as Chief Science Officer
- Researching and reports on the clinical trial processes;
- Working directly with partner companies on their development efforts
with their medications and Company Products;
- Researching drug candidates for in-licensing
- Researching disease states for Company Products.
Family Relationship Disclosure
There is no familial relationship between any of the
persons named above.
Legal Proceedings Disclosure
Marc Giroux was a named party in the Receivership
(See “Legal Proceedings and Merger” above). None of the Directors or Officers have been through a bankruptcy proceeding. None
of the Directors or Officers have any criminal convictions. None of the Directors or Officers are currently party to any civil suits as
of the Date of this Offering Circular.
COMPENSATION OF DIRECTORS AND EXECUTIVES
| Name | |
Capacities in which Compensation was Received (FY2021) | |
Cash Compensation | |
Other Compensation (Cash Value) | |
Total Compensation (FY2021) |
| Marc Giroux | |
Director/CEO | |
$ | 231,475 | | |
N/A | |
$ | 231,475 | |
| Thomas McDowell | |
Director/CFO | |
$ | 0 | | |
N/A | |
$ | 0 | |
| Glenn Cornett | |
CSO | |
$ | 0 | | |
N/A | |
$ | 0 | |
Aggregate Compensation to Directors
The Company has two (2) Directors, Total Compensation
to all Directors is $231,475
Increases in Compensation/Bonuses
The Company does intend to increase compensation
to the Directors or Officers. Upon a successful Offering, the Company intends to increase the salaries of the following persons;
Marc Giroux ($400,000 annually); Tom McDowell ($275,000 annually); Glenn Cornett ($275,000 annually). Increases of compensation to
the Officers or Directors will occur at the sole discretion of the Board.
SECURITY OWNERSHIP OF MANAGEMENT AND CERTAIN
SECURITYHOLDERS
| Title of Class | |
Name and Address of Beneficial Owner | |
Amount and nature of beneficial ownership | |
Amount and nature of beneficial ownership acquirable2 | |
Percent of Class |
| Common Stock | |
Marc Giroux 1 | |
| 43,416,595 | | |
| 96,153 | | |
| 82.94% | |
(1) 16825 48th Ave West, Suite 434, Lynnwood, WA 98037
(2) Refers to Convertible Notes held by Shareholder (see “Interest
of Management in Certain Transactions” below)
INTEREST OF MANAGEMENT IN CERTAIN TRANSACTIONS
The Company has entered into four Convertible promissory notes (“Convertible
Notes”) with the Officers of the Company. See Exhibit 8 for copies of the Convertible Notes.
Convertible Note 1:
Holder of the Convertible Note: Marc Giroux
Date of Note: May 4, 2022
Principal amount: $60,000
Interest Rate: 10% per annum
Conversion Rate: $2.08 per Share
Convertible Note 2:
Holder of the Convertible Note: Marc Giroux
Date of Note: November 6, 2021
Principal amount: $140,000
Interest Rate: 10% per annum
Conversion Rate: $2.08 per Share
Convertible Note 3:
Holder of the Convertible Note: Tom McDowell
Date of Note: September 13, 2021
Principal amount: $75,000
Interest Rate: 10% per annum
Conversion Rate: $2.08 per Share
Convertible Note 4:
Holder of the Convertible Note: Tom McDowell
Date of Note: December 4, 2021
Principal amount: $25,000
Interest Rate: 10% per annum
Conversion Rate: $2.08 per Share
Merger with Savile Thereaputics, Inc.
Prior to the Merger, Tom McDowell owned 100% of Savile Therapeutics, Inc.
Pursuant to the Merger plan, Savile shareholders received 3,011,382 Shares of Class A Common stock in the Company in exchange for the
assets of Savile. This means that Tom McDowell received all of the aforementioned Shares of Class A Common Stock in the Company. The fair
market value of the Class A Common Stock Shares at the time is difficult to determine. The par value of the Class A Common Stock at the
time was .00001 – meaning the transfer of this stock had a par value of $30.113.
DESCRIPTION OF THE SECURITIES
Summary of Company Equity
The Company has two authorized classes of equity stock,
Common Stock and Preferred Stock. The Company is authorized to issue 74,000,000 Shares of Common Stock and 1,000,000 Shares of Preferred
Stock. As of the Date of this Offering Circular, no Shares of Preferred Stock are outstanding.
Description of the Common Stock Offered through this Offering
The voting, dividend, and liquidation rights
of the holders of the Common Stock are set by the Amended and Restated Certificate of Incorporation (see Exhibit 2) and the relevant Delaware
Statutes.
Dividends, Liquidation, and Dissolution rights
Common Stock is entitled to equal rights in dividends and distributions
on cash, stock, or property when and if declared by the Board. The Common Stockholders are entitled to distributions in the event of any
liquidation, dissolution, or winding up of the Company.
Voting
The holders of the Common Stock are entitled to one
vote for each share of Common Stock held at all meetings of stockholders (and written actions in lieu of meetings). There is no cumulative
voting for Common Stock. The Common Stock has the exclusive right to vote.
Redemption Provisions
Common Stock does not have any redemption rights.
Sinking Fund Provisions
The Amended and Restated Certificate of Incorporation does not contain
any Sinking Fund Provisions.
Liability to Further Calls
The Amended and Restated Certificate of Incorporation
does not contain any provisions regarding liability to further calls.
Restrictions on Alienability
The Shares have no restrictions on alienability.
PART F/S
Financial
Statements and
Independent
Auditor’s Report
Kurve
Therapeutics, Inc.
December
31, 2021 and 2020
INDEPENDENT AUDITOR’S REPORT
To the Stockholder of Kurve Therapeutics, Inc.:
Opinion
We have audited the accompanying financial statements of Kurve Therapeutics,
Inc. (the “Company”), which comprise of the balance sheets as of December 31, 2021 and 2020, and the related statements of
operations, changes in stockholders’ equity, and cash flows for the years then ended, and the related notes to the financial statements.
In our opinion, the financial statements referred to above present fairly,
in all material respects, the financial position of the Company as of December 31, 2021 and 2020, and the results of its operations and
cash flows for the years then ended in accordance with accounting principles generally accepted in the United States of America (“U.S.
GAAP”).
Basis of Opinion
We conducted our audits in accordance with auditing standards generally
accepted in the United States of America (“U.S. GAAS”). Our responsibilities under those standards are further described in
the Auditor’s Responsibilities for the Audit of the Financial Statements section of our report. We are required to be independent
of the Company and to meet our other ethical responsibilities in accordance with the relevant ethical requirements relating to our audits.
We believe that the audit evidence we have obtained is sufficient and appropriate to provide a basis for our audit opinion.
Substantial Doubt about the Company’s Ability to Continue as a
Going Concern
The accompanying financial statements have been prepared assuming that
the Company will continue as a going concern. As discussed in Note C to the financial statements, the Company had a negative working capital
of approximately $34,000. Additionally, for the years ended December 31, 2021 and 2020, the Company incurred net losses of approximately
$65,000 and $81,000 respectively. Management’s evaluation of the events and conditions and management’s plans regarding those
matters are also described in Note C. The financial statements do not include any adjustments that might result from the outcome of this
uncertainty. Our opinion is not modified with respect to that matter.
Responsibilities of Management for the Financial Statements
Management is responsible for the preparation and fair presentation of
the financial statements in accordance with
U.S. GAAP, and for the design, implementation, and maintenance of internal
control relevant to the preparation and fair presentation of financial statements that are free from material misstatement, whether due
to fraud or error.
In preparing the financial statements, management is required to evaluate
whether there are conditions or events, considered in the aggregate, that raise substantial doubt about the Company’s ability to
continue as a going concern within one year after the date that the financial statements are available to be issued.
Auditor’s Responsibilities for the Audit of the Financial Statements
Our objectives are to obtain reasonable assurance about whether the financial
statements as a whole are free from material misstatement, whether due to fraud or error, and to issue an auditor’s report that
includes our opinion.
Reasonable assurance is a high level of assurance but is not absolute assurance and therefore is not a guarantee
that an audit conducted in accordance with U.S. GAAS will always detect a material misstatement when it exists. The risk of not detecting
a material misstatement resulting from fraud is higher than for one resulting from error, as fraud may involve collusion, forgery, intentional
omissions, misrepresentations, or the override of internal control.
Misstatements are considered material if there is a substantial likelihood
that, individually or in the aggregate, they would influence the judgment made by a reasonable user based on the financial statements.
In performing an audit in accordance with U.S. GAAS, we:
-Exercise professional judgment and maintain professional skepticism throughout
the audit.
-Identify and assess the risks of material misstatement of the financial
statements, whether due to fraud or error, and design and perform audit procedures responsive to those risks. Such procedures include
examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements.
-Obtain an understanding of internal control relevant to the audit in order
to design audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness
of the Company’s internal control. Accordingly, no such opinion is expressed.
-Evaluate the appropriateness of accounting policies used and the reasonableness
of significant accounting estimates made by management, as well as evaluate the overall presentation of the financial -statements.
-Conclude whether, in our judgment, there are conditions or events, considered
in the aggregate, that raise substantial doubt about the Company’s ability to continue as a going concern for a reasonable period
of time.
We are required to communicate with those charged with governance regarding,
among other matters, the planned scope and timing of the audit, significant audit findings, and certain internal control related matters
that we identified during the audit.
s/Assurance Dimensions
Jacksonville, Florida
February 11, 2022
Kurve Therapeutics,
Inc.
Balance Sheets
As of December
31, 2021 and 2020
| ASSETS | |
2021 | |
2020 |
| Current assets: | |
| | | |
| | |
| Cash | |
$ | 373,804 | | |
$ | 46,300 | |
| Accounts receivable | |
| — | | |
| 3,821 | |
| Due from related party | |
| — | | |
| 36,905 | |
| Note receivable - related party | |
| 15,000 | | |
| — | |
| Prepaid expenses | |
| 251,770 | | |
| — | |
| TOTAL ASSETS | |
$ | 640,574 | | |
$ | 87,026 | |
| LIABILITIES AND STOCKHOLDERS’ EQUITY | |
| | | |
| | |
| Current liabilities: | |
| | | |
| | |
| Accounts payable | |
$ | 2,870 | | |
$ | 1,844 | |
| Accrued expenses | |
| 25,461 | | |
| 9,657 | |
| Convertible notes payable | |
| 507,500 | | |
| — | |
| Deferred revenue | |
| 52,500 | | |
| 26,250 | |
| Due to related party | |
| 18,395 | | |
| — | |
| TOTAL LIABILITIES | |
| 606,726 | | |
| 37,751 | |
| Stockholders’ equity: | |
| | | |
| | |
| Preferred stock; $0.001 par value; 1,000,000 shares authorized, and no shares issued and outstanding | |
| — | | |
| — | |
| Common stock; $0.001 par value; 74,000,000 shares authorized, and 50,189,694 founder shares issued and outstanding, and 118,461 and 94,423 shares issued and outstanding, respectively | |
| 118 | | |
| 94 | |
| Additional paid-in capital | |
| 179,882 | | |
| 129,906 | |
| Accumulated deficit | |
| (146,152) | | |
| (80,725) | |
| TOTAL STOCKHOLDERS’ EQUITY | |
| 33,848 | | |
| 49,275 | |
| TOTAL LIABILITIES AND STOCKHOLDERS’ EQUITY | |
$ | 640,574 | | |
$ | 87,026 | |
Kurve Therapeutics,
Inc.
Statements
of Operations
For the Years
Ended December 31, 2021 and 2020
| | |
2021 | |
2020 |
| REVENUE | |
$ | 70,700 | | |
$ | 38,815 | |
| OPERATING EXPENSES: | |
| | | |
| | |
| Wages & benefits | |
| 87,158 | | |
| 28,918 | |
| Sales & marketing | |
| 25,375 | | |
| 17,794 | |
| Research & development | |
| 7,254 | | |
| 60,098 | |
| Office expenses | |
| 3,636 | | |
| 2,487 | |
| Bad debt expense | |
| 1,921 | | |
| — | |
| Conferences, travel & entertainment | |
| 1,870 | | |
| 7,290 | |
| Professional fees | |
| 630 | | |
| 1,765 | |
| Computer expenses | |
| 500 | | |
| 1,188 | |
| Total operating expenses | |
| 128,344 | | |
| 119,540 | |
| Net operating loss | |
| (57,644 | ) | |
| (80,725 | ) |
| OTHER EXPENSES: | |
| | | |
| | |
| Interest expense | |
| (7,783 | ) | |
| — | |
| NET LOSS | |
$ | (65,427 | ) | |
$ | (80,725 | ) |
Kurve
Therapeutics, Inc.
Statements
of Changes in Stockholders’ Equity
For the Years
Ended December 31, 2021 and 2020
| | |
Preferred Stock | |
| |
Common Stock | |
| |
| |
| |
|
| | |
Shares | |
Amount | |
Shares | |
Amount | |
Additional Paid-In Capital | |
Accumulated Deficit | |
Total |
| January 1, 2020 | |
| — | | |
$ | — | | |
| — | | |
$ | — | | |
$ | — | | |
$ | — | | |
$ | — | |
| Issuance of common stock - founders’ shares | |
| — | | |
| — | | |
| 50,189,694 | | |
| — | | |
| — | | |
| — | | |
| — | |
| Issuance of common stock | |
| — | | |
| — | | |
| 94,423 | | |
| 94 | | |
| 129,906 | | |
| — | | |
| 130,000 | |
| Net loss | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| (80,725 | ) | |
| (80,725 | ) |
| December 31, 2020 | |
| — | | |
| — | | |
| 50,284,117 | | |
| 94 | | |
| 129,906 | | |
| (80,725 | ) | |
| 49,275 | |
| Issuance of common stock | |
| — | | |
| — | | |
| 24,038 | | |
| 24 | | |
| 49,976 | | |
| — | | |
| 50,000 | |
| Net loss | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| (65,427 | ) | |
| (65,427 | ) |
| December 31, 2021 | |
| — | | |
$ | — | | |
| 50,308,155 | | |
$ | 118 | | |
$ | 179,882 | | |
$ | (146,152 | ) | |
$ | 33,848 | |
Kurve Therapeutics,
Inc.
Statements
of Cash Flows
For the Years
Ended December 31, 2021 and 2020
| | |
2021 | |
2020 |
| CASH FLOWS FROM OPERATING ACTIVITIES: | |
| | | |
| | |
| Net loss | |
$ | (65,427 | ) | |
$ | (80,725 | ) |
| Adjustments to reconcile net loss to net cash provided (used) by operating activities: | |
| | | |
| | |
| Accounts receivable | |
| 3,821 | | |
| (3,821 | ) |
| Prepaid expenses | |
| (3,000 | ) | |
| — | |
| Deferred revenue | |
| 26,250 | | |
| 26,250 | |
| Due to/(from) related party | |
| 55,300 | | |
| (36,905 | ) |
| Accounts payable and accrued expenses | |
| 16,830 | | |
| 11,501 | |
| Net cash provided (used) by operating activities | |
| 33,774 | | |
| (83,700 | ) |
| CASH FLOWS FROM INVESTING ACTIVITIES: | |
| | | |
| | |
| Loan to related party | |
| (15,000 | ) | |
| — | |
| Net cash used by investing activities | |
| (15,000 | ) | |
| — | |
| CASH FLOWS FROM FINANCING ACTIVITIES: | |
| | | |
| | |
| Prepaid equity raise costs | |
| (248,770 | ) | |
| — | |
| Proceeds from issuance of common stock | |
| 50,000 | | |
| 130,000 | |
| Proceeds from convertible notes payable | |
| 507,500 | | |
| — | |
| Net cash provided by financing activities | |
| 308,730 | | |
| 130,000 | |
| NET CHANGE IN CASH | |
| 327,504 | | |
| 46,300 | |
| Cash at beginning of year | |
| 46,300 | | |
| — | |
| Cash at end of year | |
$ | 373,804 | | |
$ | 46,300 | |
| SUPPLEMENTAL DISCLOSURE OF CASH FLOW INFORMATION | |
| | | |
| | |
| Cash paid during year for interest | |
$ | — | | |
$ | — | |
| Cash paid during year for income taxes | |
$ | — | | |
$ | — | |
Note A – Nature of Business
Kurve Therapeutics, Inc. (the “Company”) is a private company
incorporated in the state of Delaware. The Company is developing disease-modifying drugs with demonstrated clinical efficacy in cognitive
aging, neurodegenerative diseases and related disorders, and develops nasal drug delivery devices.
Note B – Summary of Significant Accounting Policies
Basis of Accounting
The Company reports the results of its operations
using the accrual method of accounting for financial statement purposes. Under this method, income is recognized when earned and expenses
are deducted when incurred. The accounting policies of the Company are in accordance with generally accepted accounting principles in
the United States of America (“U.S. GAAP”).
Use of Estimates
The preparation of financial statements in conformity
with U.S. GAAP requires management to make estimates and assumptions that affect the amounts reported of assets and liabilities and disclosure
of contingent assets and liabilities at the date of the consolidated financial statements and the reported amounts of revenues and expenses
during the reporting period. Actual results could differ from those estimates.
Recently Adopted Accounting Standards
Accounting standards promulgated by the Financial
Accounting Standards Board (“FASB”) are subject to change. Changes in such standards may have an impact on the Company’s
future financial statements.
The Company periodically reviews new accounting standards
that are issued. Although some of these accounting standards may be applicable to the Company, the Company has identified the following
new accounting standard that it believes merits further discussion.
Recent Accounting Pronouncements Not Yet Adopted
In August 2020, the FASB issued Accounting Standards
Update (“ASU”) 2020-06, Accounting for Convertible Instruments and Contracts in an Entity’s Own Equity, as part of its
overall simplification initiative to reduce costs and complexity of applying accounting standards while maintaining or improving the usefulness
of the information provided to users of financial statements. Among other changes, the new guidance removes from U.S. GAAP separation
models for convertible debt that require the convertible debt to be separated into a debt and equity component, unless the conversion
feature is required to be bifurcated and accounted for as a derivative or the debt is issued at a substantial premium. As a result, after
adopting the guidance, entities will no longer separately present such embedded conversion features in equity and will instead account
for the convertible debt wholly as debt. The guidance is effective for financial statements issued for fiscal years beginning after December
15, 2021. Management is evaluating the impact of this ASU on the Company’s financial reporting.
Other accounting standards that have been issued or
proposed by FASB that do not require adoption until a future date are not expected to have a material impact on the financial statements
upon adoption.
Cash
The Company deposits cash and cash equivalents
with financial institutions which management believes is of high credit quality. The Company does not believe it is exposed to
significant risk. At December 31, 2021 and 2020, there were no cash equivalents.
Convertible Notes Payable
The Company evaluates all conversion and redemption
features contained in a debt instrument to determine if there are any embedded features that require bifurcation as a derivative or separation
as a beneficial conversion feature. Based on management’s review of the convertible notes, no bifurcation of the embedded features
was considered necessary as of December 31, 2021 and 2020.
Revenue Recognition
All revenues from exchange transactions are recorded
in accordance with ASC 606 which is recognized when: (i) a contract with a customer has been identified, (ii) the performance obligation(s)
in the contract have been identified, (iii) the transaction price has been determined, (iv) the transaction price has been allocated to
each performance obligation in the contract, and (v) the Company has satisfied the applicable performance obligation at a point in time
or over time.
The Company acts as an agent to secure customer contracts
for the purchase of medical devices developed and manufactured by Kurve Technology, Inc. (“Kurve Technology”), a related party
through common ownership. The Company earns a commission when the devices are delivered to the customer. All payments for medical devices
are due prior to shipment of the product.
Costs of goods sold include all direct materials related
to contract performance. General and administrative costs are charged to expense as incurred.
The contract liabilities as of December 31, 2021 and
2020 were composed of deferred revenue related to customer deposits on orders that have not shipped.
Research & Development
The Company expenses product testing and research
& development costs as incurred. Research and development costs are primarily composed of clinical trial costs, manufacturing costs
for both clinical and pre-clinical materials as well as other contracted services, and other external costs. Nonrefundable advance payments
for goods and services that will be used in future research and development activities are expensed when the activity is performed or
when the goods have been received, rather than when payment is made, in accordance with ASC 730, Research and Development.
Income Taxes
The Company utilizes an asset and liability approach
to financial accounting and reporting for income taxes. Deferred income tax assets and liabilities are computed annually for differences
between the financial statement and tax basis of assets and liabilities that will result in taxable deductible amounts in the future based
on enacted tax laws and rates applicable to the periods in which the differences are expected to affect taxable income. Valuation allowances
are established when necessary to reduce deferred tax assets to the amount expected to be realized.
The Company’s income tax returns are subject
to review and examination by federal and state authorities in accordance with prescribed statutes. When accrued, interest and penalties
related to unpaid taxes are included in income tax expense.
The Company adopted the income tax standard for uncertain
tax positions. As a result of this implementation, the Company evaluated its tax positions and determined that it has no uncertain tax
positions as of December 31, 2021. The Company’s 2020 and 2021 tax years are open for examination for federal and state taxing authorities.
Fair Value Measurements
The carrying value of cash, accounts receivable, accounts
payable and accrued liabilities approximates fair value due to the short terms to maturity of these instruments. The carrying value of
the note receivable – related party and notes payable would not differ significantly if recalculated based on current interest rates.
No financial instruments are held for trading purposes.
Note C – Going Concern
The accompanying financial statements have been prepared
assuming the Company will continue as a going concern. At December 31, 2021, the Company had a negative working capital of approximately
$34,000. Additionally, for the years ended December 31, 2021 and 2020, the Company incurred net losses of approximately $65,000 and $81,000
respectively. These matters raise doubt about the Company's ability to continue as a going concern. The ability of the Company to continue
as a going concern is dependent upon increasing sales and obtaining additional capital and financing.
Management plans to sell shares of common stock through
private and public offerings. While the Company believes in the viability of its ability to raise additional funds, there can be no assurances
to that effect. The financial statements do not include adjustments to reflect the possible effects on the recoverability and classification
of assets or the amounts and classification of liabilities that may result from the outcome of this uncertainty.
Note D – Note Receivable – Related
Party
In November 2021, the Company entered into a promissory
note agreement with Savile Therapeutics, Inc. (“Savile”), a related party through common ownership. The promissory note allows
for borrowings of up to $405,000, bears interest at an annual rate of 10% and is due and payable on November 1, 2022. As of December 31,
2021, $15,000 had been loaned to Savile. In January 2022, the Company disbursed an additional $390,000 to Savile.
Savile has made an offer to purchase Kurve Technology,
which is under the direction of a General Receiver per revised Code of Washington (State), Chapter 7.60. Savile has made an offer to purchase
Kurve Technology’s assets for $5,000,000, and has made a deposit of $405,000 with the Receiver. The balance of the purchase price
will be a note payable accruing interest at 5% per annum and due in full on or before two years from the date of the closing. Savile also
agrees to pay a royalty of 5% of gross revenue to the Receiver for distribution to the stockholders of Kurve Technology for the first
five years following the sale of its first product. The Receiver has approved the transaction to be presented to the Snohomish County
Court. The court’s decision is expected in early March 2022.
Note E – Convertible Notes Payable
During September through December 2021, the
Company entered into various convertible promissory note agreements with various holders. The note agreements range from $5,000 -
$140,000, bear interest at 10% are due and payable between September 2022 and December 2022. The notes payable provide a conversion
option at the sole discretion of the holder to convert the unpaid principal balance, plus accrued interest for common stock at a
purchase price of $2.08 per share.
Note F – Related Party Transactions
Kurve Technology has completed two studies through
Phase 2 including; Mild Cognitive Impairment (“MCI”) in Alzheimer’s (Wake Forest) and MCI in Diabetes (Harvard Medical
School). Results of these studies have been presented at the meeting of the Alzheimer’s Association and America Diabetes Association.
In addition, Kurve Technology has developed technology to successfully deliver drugs from the nose to the brain for central nervous system
disorders at any level.
The Company acts as an agent for the sale of medical
devices developed and manufactured by Kurve Technology. The Company earns a 35% commission on all medical devices for contracts that it
secures. As of December 31, 2021, the Company has completed agreements and commenced clinical testing with nine organizations. As of December
31, 2021, there was a net due to Kurve Technology of $18,395. As of December 31, 2020, there was a net due from Kurve Technology of $36,905.
Note G – Income Taxes
No provision or benefit for federal or state income
taxes has been reflected for the years ended December 31, 2021 and 2020 since the Company reported losses and has established a valuation
allowance against the total net deferred tax asset.
Significant components of the Company’s deferred
tax assets (liabilities) as of December 31:
| | |
2021 | |
2020 |
| Net operating loss carryforward | |
$ | 26,104 | | |
$ | 14,409 | |
| Valuation allowance | |
| (26,104) | | |
| (14,409) | |
| Net deferred tax asset | |
$ | — | | |
$ | — | |
The entire balance of the deferred tax asset has been
offset by a valuation allowance since the utilization of net operating loss carryforwards cannot be sufficiently assured at December 31,
2021 and 2020.
At December 31, 2021 and 2020, the Company had federal
net operating loss carryforwards of approximately $116,000 and $64,000, respectively. The federal net operating loss may be carried forward
indefinitely but are limited to 80% of the taxable income in any one tax period. The utilization of the net operating loss carryforwards
may be limited in the case of certain events including significant changes in ownership interests. The Company’s federal and state
blended tax rate was 21% for the years ended December 31, 2021 and 2020.
Note H – Stock Warrants
The Company issued warrants for the purchase of 80,000
shares of common stock at an exercise price of $1.25 per share. The warrants are fully vested and expire between December 8, 2029 and
January 3, 2030. During the years ended December 31, 2021 and 2020, no warrants were exercised.
Note I – Concentrations and Commitments
COVID-19
Due to the COVID-19 outbreak in 2020, the Company
was not closed at any point. Additionally, there were no staff furloughed or terminated due to COVID-19. Overall, the Company was not
significantly affected by COVID-19.
Due to the level of risk this virus may have on the
global economy, it is at least reasonably possible that it could have an impact on the operations of the Company in the near term that
could materially impact the Company’s financial statements, however, management does not believe there will be any future impact.
In addition, with vaccines and therapeutics coming to market, these will also help to mitigate any potential future losses.
Note J – Subsequent Events
In preparing the accompanying financial statements,
the Company has evaluated events and transactions for potential recognition or disclosure through February 11, 2022, the date the financial
statements were available to be issued. Management has determined that all subsequent events have been properly disclosed.
Financial
Statements and
Independent
Auditor’s Report
Kurve
Technology, Inc.
December
31, 2021 and 2020
INDEPENDENT
AUDITOR’S REPORT
To the Stockholders
of Kurve Technology, Inc.:
Opinion
We have audited
the accompanying financial statements of Kurve Technology, Inc. (the “Company”), which comprise of the balance sheets as
of December 31, 2021 and 2020, and the related statements of operations, changes in stockholders’ deficit, and cash flows for the
years then ended, and the related notes to the financial statements.
In our opinion,
the financial statements referred to above present fairly, in all material respects, the financial position of the Company as of December
31, 2021 and 2020, and the results of its operations and cash flows for the years then ended in accordance with accounting principles
generally accepted in the United States of America (“U.S. GAAP”).
Basis of Opinion
We conducted
our audits in accordance with auditing standards generally accepted in the United States of America (“U.S. GAAS”). Our responsibilities
under those standards are further described in the Auditor’s Responsibilities for the Audit of the Financial Statements section
of our report. We are required to be independent of the Company and to meet our other ethical responsibilities in accordance with the
relevant ethical requirements relating to our audits. We believe that the audit evidence we have obtained is sufficient and appropriate
to provide a basis for our audit opinion.
Substantial
Doubt about the Company’s Ability to Continue as a Going Concern
The accompanying
financial statements have been prepared assuming that the Company will continue as a going concern. As discussed in Note B to the financial
statements, the Company is in receivership. Additionally as discussed in Note C, the Company had a negative working capital of approximately
$5,002,000 and $4,641,000 as of December 31, 2021 and 2020, respectively. For the years ended December 31, 2021 and 2020, the Company
incurred net losses of approximately $385,000 and $417,000, respectively. And the Company had negative cash flows from operations in
the amounts of approximately $16,000 and $14,000, respectively. Management’s evaluation of the events and conditions and management’s
plans regarding those matters are also described in Note C. The financial statements do not include any adjustments that might result
from the outcome of this uncertainty. Our opinion is not modified with respect to that matter.
Responsibilities
of Management for the Financial Statements
Management is
responsible for the preparation and fair presentation of the financial statements in accordance with U.S. GAAP, and for the design, implementation,
and maintenance of internal control relevant to the preparation and fair presentation of financial statements that are free from material
misstatement, whether due to fraud or error.
In preparing
the financial statements, management is required to evaluate whether there are conditions or events, considered in the aggregate, that
raise substantial doubt about the Company’s ability to continue as a going concern within one year after the date that the financial
statements are available to be issued.
Auditor’s
Responsibilities for the Audit of the Financial Statements
Our objectives
are to obtain reasonable assurance about whether the financial statements as a whole are free from material misstatement, whether due
to fraud or error, and to issue an auditor’s report that includes our opinion.
Reasonable assurance
is a high level of assurance but is not absolute assurance and therefore is not a guarantee that an audit conducted in accordance with
U.S. GAAS will always detect a material misstatement when it exists. The risk of not detecting a material misstatement resulting from
fraud is higher than for one resulting from error, as fraud may involve collusion, forgery intentional omissions, misrepresentations,
or the override of internal control. Misstatements are considered material if there is a substantial likelihood that, individually or
in the aggregate, they would influence the judgement made by a reasonable user based on the financial statements.
In performing
an audit in accordance with U.S. GAAS, we:
·
Exercise professional judgement and maintain professional skepticism throughout the audit.
·
Identify and assess the risks of material misstatement of the financial statements, whether due to fraud or error, and design and perform
audit procedures responsive to those risks. Such procedures include examining, on a test basis, evidence regarding the amounts and disclosures
in the financial statements.
·
Obtain an understanding of internal control relevant to the audit in order to design audit procedures that are appropriate in the circumstances,
but not for the purpose of expressing an option on the effectiveness of the Company’s internal control. Accordingly, no such opinion
is expressed.
·
Evaluate the appropriateness of accounting policies used and the reasonableness of significant accounting estimates made by management,
as well as evaluate the overall presentation of the financial statements.
·
Conclude whether, in our judgement, there are conditions or events considered in the aggregate, that raise substantial doubt about the
Company’s ability to continue as a going concern for a reasonable period of time.
We are required
to communicate with those charged with governance regarding, among other matters, the planned scope and timing of the audit, significant
audit findings, and certain internal control related matters that we identified during the audit.
s//: Assurance
Dimensions
Jacksonville,
Florida
February 11,
2022
Kurve Technology,
Inc.
Balance Sheets
As of December
31, 2021 and 2020
| ASSETS | |
2021 | |
2020 |
| Current assets: | |
| | | |
| | |
| Cash | |
$ | 16,355 | | |
$ | 32,662 | |
| Accounts receivable, net | |
| 6,000 | | |
| 12,350 | |
| Due from related party | |
| 18,395 | | |
| — | |
| Total current assets | |
| 40,750 | | |
| 45,012 | |
| Intangible assets: | |
| | | |
| | |
| Patents, net | |
| 55,230 | | |
| 78,437 | |
| Total intangible assets | |
| 55,230 | | |
| 78,437 | |
| TOTAL ASSETS | |
$ | 95,980 | | |
$ | 123,449 | |
| LIABILITIES AND STOCKHOLDERS’ DEFICIT | |
| | | |
| | |
| Current liabilities: | |
| | | |
| | |
| Accounts payable | |
$ | 393,543 | | |
$ | 374,464 | |
| Accrued expenses | |
| 2,905,529 | | |
| 2,578,876 | |
| Deferred revenue | |
| 97,500 | | |
| 48,750 | |
| Notes payable | |
| 1,646,616 | | |
| 1,646,616 | |
| Due to related party | |
| — | | |
| 36,905 | |
| TOTAL LIABILITIES | |
| 5,043,188 | | |
| 4,685,611 | |
| Stockholders’ deficit: | |
| | | |
| | |
| Preferred stock - par value $0.001, 1,000,000 shares authorized, and 500,000 shares issued and outstanding | |
| 500 | | |
| 500 | |
| Common stock - par value $0.001, 50,000,000 shares authorized, and 29,095,456 shares issued and outstanding | |
| 29,095 | | |
| 29,095 | |
| Additional paid-in capital | |
| 6,320,889 | | |
| 6,320,889 | |
| Accumulated deficit | |
| (11,297,692 | ) | |
| (10,912,646 | ) |
| TOTAL STOCKHOLDERS’ DEFICIT | |
| (4,947,208 | ) | |
| (4,562,162 | ) |
| TOTAL LIABILITIES AND STOCKHOLDERS’ DEFICIT | |
$ | 95,980 | | |
$ | 123,449 | |
Kurve Technology,
Inc.
Statements
of Operations
For the Years
Ended December 31, 2021 and 2020
| | |
2021 | |
2020 |
| REVENUE | |
$ | 288,890 | | |
$ | 312,685 | |
| OPERATING EXPENSES: | |
| | | |
| | |
| Wages & benefits | |
| 144,317 | | |
| 157,702 | |
| Research & development | |
| 118,971 | | |
| 141,618 | |
| Professional fees | |
| 60,630 | | |
| 70,490 | |
| Rent expense | |
| 25,347 | | |
| 25,272 | |
| Office expenses | |
| 25,021 | | |
| 27,920 | |
| Patent amortization | |
| 23,207 | | |
| 47,350 | |
| Computer expenses | |
| 7,771 | | |
| 5,139 | |
| Travel expenses | |
| 905 | | |
| 3,679 | |
| Marketing expenses | |
| 602 | | |
| 526 | |
| Bad debt expanse | |
| — | | |
| 6,550 | |
| Total operating expenses | |
| 406,771 | | |
| 486,246 | |
| NET OPERATING LOSS | |
| (117,881 | ) | |
| (173,561 | ) |
| OTHER EXPENSES: | |
| | | |
| | |
| Interest expense | |
| (267,165 | ) | |
| (243,112 | ) |
| NET LOSS | |
$ | (385,046 | ) | |
$ | (416,673 | ) |
Kurve Technology,
Inc.
Statements
of Changes in Stockholders’ Deficit
For the Years
Ended December 31, 2021 and 2020
| | |
Preferred Stock | |
| |
Common Stock | |
| |
| |
| |
|
| | |
Shares | |
Amount | |
Shares | |
Amount | |
Additional Paid-In Capital | |
Accumulated Deficit | |
Total |
| Balance December 31, 2019 | |
| 500,000 | | |
$ | 500 | | |
| 29,095,456 | | |
$ | 29,095 | | |
$ | 6,320,889 | | |
$ | (10,495,973 | ) | |
$ | (4,145,489 | ) |
| Net loss | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| (416,673 | ) | |
| (416,673 | ) |
| December 31, 2020 | |
| 500,000 | | |
| 500 | | |
| 29,095,456 | | |
| 29,095 | | |
| 6,320,889 | | |
| (10,912,646 | ) | |
| (4,562,162 | ) |
| Net loss | |
| — | | |
| — | | |
| — | | |
| — | | |
| — | | |
| (385,046 | ) | |
| (385,046 | ) |
| December 31, 2021 | |
| 500,000 | | |
$ | 500 | | |
| 29,095,456 | | |
$ | 29,095 | | |
$ | 6,320,889 | | |
$ | (11,297,692 | ) | |
$ | (4,947,208 | ) |
Kurve Technology,
Inc.
Statements
of Cash Flows
For the Years
Ended December 31, 2021 and 2020
| | |
2021 | |
2020 |
| CASH FLOWS FROM OPERATING ACTIVITIES: | |
| | | |
| | |
| Net loss | |
$ | (385,046 | ) | |
$ | (416,673 | ) |
| Adjustments to reconcile net loss to net cash used by operating activities: | |
| | | |
| | |
| Patent amortization | |
| 23,207 | | |
| 47,350 | |
| Bad debt expense | |
| — | | |
| 6,550 | |
| Change in cash due to changes in: | |
| | | |
| | |
| Accounts receivable, net | |
| 6,350 | | |
| (11,500 | ) |
| Accounts payable and accrued expenses | |
| 345,732 | | |
| 299,663 | |
| Deferred revenue | |
| 48,750 | | |
| 48,750 | |
| Due to/(from) related party | |
| (55,300 | ) | |
| 11,905 | |
| Net cash used by operating activities | |
| (16,307 | ) | |
| (13,955 | ) |
| NET CHANGE IN CASH | |
| (16,307 | ) | |
| (13,955 | ) |
| Cash at beginning of year | |
| 32,662 | | |
| 46,617 | |
| Cash at end of year | |
$ | 16,355 | | |
$ | 32,662 | |
| SUPPLEMENTAL DISCLOSURE OF CASH FLOW INFORMATION: | |
| | | |
| | |
| Cash paid during year for interest | |
$ | — | | |
$ | — | |
| Cash paid during year for income taxes | |
$ | — | | |
$ | — | |
Kurve Technology,
Inc.
Notes to the
Financial Statements
December 31,
2021 and 2020
Note A –
Nature of Business
Kurve Technology,
Inc. (the “Company”) was organized in 2002 in the State of Delaware, and is headquartered in Lynnwood, Washington. The Company
has developed disease-modifying technology to deliver selected drugs directly from the nose to the brain.
The Company has
completed two studies through Phase 2 including; Mild Cognitive Impairment (“MCI”) in Alzheimer’s (Wake Forest) and
MCI in Diabetes (Harvard Medical School). Results of these studies have been presented at the meeting of the Alzheimer’s Association
and America Diabetes Association. In addition, the Company has developed technology to successfully deliver drugs from the nose to the
brain for central nervous system disorders at any level. Furthermore, the Company has commenced clinical testing nine organizations.
Note B –
Summary of Significant Accounting Policies
Basis of Accounting
The Company reports
the results of its operations using the accrual method of accounting for financial statement purposes. Under this method, income is recognized
when earned and expenses are deducted when incurred. The accounting policies of the Company are in accordance with generally accepted
accounting principles in the United States of America (“U.S. GAAP”).
Receivership
Under the Revised
Code of Washington (“RCW”), Chapter 7.60, the Company is under the direction of a General Receiver. Pursuant to RCW 7.60.260,
a general receiver is able to sell assets of the company over which it has been appointed free and clear of liens. This is similar to
section 362 of the bankruptcy code.
The Company desires
to exist receivership either through a sale of assets or additional investors. As such, the Company is under a non-binding letter of
intent with a related entity due to common ownership to sell select assets to raise sufficient capital to retire all of the Company’s
debt. The related entity has made an officer to purchase Kurve Techology’s assets for $5,000,000, and has made a deposit of $405,000
with the Receiver. The balance of the purchase price will be a note payable accruing interest at 5% per annum and due in full on or before
two years from the date of the closing. The related entity also agrees to pay a royalty of 5% of gross revenue to the Receiver for distribution
to the stockholders of Kurve Technology for the first five years following the sale of its first product. The Receiver has approved the
transaction to be presented to the Snohomish County Court. The court’s decision is expected in early March 2022.
As of December
31, 2021 and 2020, the General Receiver was due approximately $600,000 and $540,000 for unpaid services.
Use of Estimates
The preparation
of financial statements in conformity with U.S. GAAP requires management to make estimates and assumptions that affect the amounts reported
of assets and liabilities and disclosure of contingent assets and liabilities at the date of the consolidated financial statements and
the reported amounts of revenues and expenses during the reporting period. Actual results could differ from those estimates.
Recently Adopted
Accounting Standards
Accounting standards
promulgated by the Financial Accounting Standards Board (“FASB”) are subject to change. Changes in such standards may have
an impact on the Company’s future financial statements.
The Company periodically
reviews new accounting standards that are issued. Although some of these accounting standards may be applicable to the Company, the Company
has identified the following new accounting standard that it believes merits further discussion.
Note B –
Summary of Significant Accounting Policies (continued)
The Company adopted
Accounting Standards Codification (“ASC”) 606, Revenue from Contracts with Customers, as of January 1, 2019 using
the modified retrospective method for all contracts with customers. The adoption did not result is an impact on opening retained earnings.
No significant judgements were made in the application of the guidance in ASC 606.
Recent Accounting
Pronouncements Not Yet Adopted
In February 2016,
the FASB issued Accounting Standards Update (“ASU”) 2016-02, Leases (Topic 842). This guidance amends the existing
accounting standards for lease accounting, including requiring lessees to recognize most leases on their balance sheet. It also makes
targeted changes to lessor accounting, including a change to the treatment of initial direct leasing costs, which no longer consider
fixed internal leasing salaries as capitalized costs. Under ASU 2020-05, ASU 2016-02 is effective for years beginning on or after December
15, 2021. Management is evaluating the impact on this ASU on the Company’s financial reporting.
In August 2020,
the FASB issued ASU 2020-06, Accounting for Convertible Instruments and Contracts in an Entity’s Own Equity, as part of
its overall simplification initiative to reduce costs and complexity of applying accounting standards while maintaining or improving
the usefulness of the information provided to users of financial statements. Among other changes, the new guidance removes from GAAP
separation models for convertible debt that require the convertible debt to the separated into a debt and equity component, unless the
conversion feature is required to be bifurcated and accounted for as a derivative or the debt is issued at a substantial premium. As
a result, after adopting the guidance, entities will no longer separately present such embedded conversion features in equity and will
instead accounting for the convertible debt wholly as debt. The guidance is effective for financial statements issued for fiscal years
beginning after
December 15, 2021. Management is evaluating the impact on this ASU on the Company’s financial reporting.
Other accounting
standards that have been issued or proposed by FASB that do not require adoption until a future date are not expected to have a material
impact on the financial statements upon adoption.
Cash
The Company deposits
cash and cash equivalents with financial institutions which management believes is of high credit quality. The Company does not believe
it is exposed to significant risk. At December 31, 2021 and 2020, there were no cash equivalents.
Intangible
Assets
Intangible assets
consist of patents and patents pending. Patents and patents pending are being amortized on the straight-line method over fifteen years,
but not exceeding the initial expiration date of the patent.
Revenue Recognition
All revenues
from exchange transactions are recorded in accordance with ASC 606 which is recognized when: (i) a contract with a customer has been
identified, (ii) the performance obligation(s) in the contract have been identified, (iii) the transaction price has been determined,
(iv) the transaction price has been allocated to each performance obligation in the contract, and (v) the Company has satisfied the applicable
performance obligation at a point in time or over time.
The Company’s
revenues are primarily generated from the sale of medical devices to customers. Sales contain a single delivery element and revenue for
such sales is recognized when the customer obtains control, which is typically when the product is delivered. All payments for medical
devices are due prior to shipment of the product.
Costs of goods
sold include all direct materials related to contract performance. General and administrative costs are charged to expense as incurred.
The contract
liabilities as of December 31, 2021 and 2020 were composed of deferred revenue related to customer deposits on orders that have not shipped.
Note B –
Summary of Significant Accounting Policies (continued)
Research &
Development
The Company expenses
product testing and research & development costs as incurred. Research and development costs are primarily composed of clinical trial
costs, manufacturing costs for both clinical and pre-clinical materials as well as other contracted services, and other external
costs. Nonrefundable
advance payments for goods and services that will be used in future research and development activities are expensed when the activity
is performed or when the goods have been received, rather than when payment is made, in accordance with ASC 730, Research and Development.
Income Taxes
The Company utilizes
an asset and liability approach to financial accounting and reporting for income taxes. Deferred income tax assets and liabilities are
computed annually for differences between the financial statement and tax basis of assets and liabilities that will result in taxable
deductible amounts in the future based on enacted tax laws and rates applicable to the periods in which the differences are expected
to affect taxable income. Valuation allowances are established when necessary to reduce deferred tax assets to the amount expected to
be realized.
The Company’s
income tax returns are subject to review and examination by federal and state authorities in accordance with prescribed statutes. When
accrued, interests and penalties related to unpaid taxes are included in income tax expense.
The Company adopted
the income tax standard for uncertain tax positions. As a result of this implementation, the Company evaluated its tax positions and
determined that it has no uncertain tax positions as of December 31, 2021. The Company’s 2020 and 2021 tax years are open for examination
for federal and state taxing authorities.
Fair Value
Measurements
The carrying
value of cash, accounts receivable, accounts payable and accrued liabilities approximates fair value due to the short terms to maturity
of these instruments. The carrying value of the note receivable – related party and notes payable would not differ significantly
if recalculated based on current interest rates. No financial instruments are held for trading purposes.
Note C –
Going Concern
The accompanying
financial statements have been prepared assuming the Company will continue as a going concern, albeit under receivership. As of December
31, 2021 and 2020, the Company had an accumulated deficit of approximately $11,298,000 and $10,913,000, and a negative working capital
of approximately $5,002,000 and $4,641,000. Additionally, for the years ended December 31, 2021 and 2020, the Company incurred net losses
of approximately $385,000 and $417,000, respectively. Further, the Company has had negative cash flows from operations in amounts of
approximately $16,000 and $14,000, respectively. These matters raise substantial doubt about the Company’s ability to continue
as a going concern. The ability of the Company to continue as a going concern is dependent upon increasing sales and obtaining additional
capital and financing.
The Company has
entered an agreement to sell its assets to the related entity as described in Note B, which is designed to retire all of the Company’s
debt. While the Company believes in the viability of this transaction, there can be no assurances to that effect. The financial statements
do not include adjustments to reflect the possible effects on the recoverability and classification of assets or the amounts and classification
of liabilities that may result from the outcome of this uncertainty.
Note D –
Intangible Assets
Intangible assets
consisted of the following as of December 31:
| | |
2021 | |
2020 | |
Useful Life (years) |
| Patents | |
$ | 456,241 | | |
$ | 456,241 | | |
| 15 | |
| Trademarks | |
| 12,053 | | |
| 12,053 | | |
| indefinite | |
| Total intangible assets | |
| 468,294 | | |
| 468,294 | | |
| | |
| Less: accumulated amortization | |
| (413,064 | ) | |
| (389,857 | ) | |
| | |
| Intangible assets, net | |
$ | 55,230 | | |
$ | 78,437 | | |
| | |
Amortization
expense charged to operations for the years ended December 31, 2021 and 2020 was approximately $23,000 and $47,000, respectively.
The following
is a schedule of the estimated amortization expense for intangible assets over the remaining useful life:
| Years ending December 31, | |
|
| | 2022 | | |
$ | 22,008 | |
| | 2023 | | |
| 15,952 | |
| | 2024 | | |
| 11,680 | |
| | 2025 | | |
| 5,590 | |
| | | | |
$ | 55,230 | |
Note E –
Accrued Expenses
Accrued liabilities
consisted of the following as of December 31:
| | |
2021 | |
2020 |
| Accrued interest | |
$ | 1,574,894 | | |
$ | 1,307,728 | |
| Accrued wages and benefits | |
| 729,705 | | |
| 729,705 | |
| Accrued fees due to the General Receiver | |
| 600,111 | | |
| 540,006 | |
| Other | |
| 819 | | |
| 1,437 | |
| | |
$ | 2,905,529 | | |
$ | 2,578,876 | |
Note F –
Notes Payable
The Company carries
notes payable due to multiple lenders. All notes have an interest rate of 10% and have matured. Interest accrues each month. Selected
lenders also received common stock warrants with per share exercise prices ranging from $0.64 to $1.00. Unexpired common stock warrants
are presented in Note I. The balance of the notes payable as of December 31:
| | |
2021 | |
2020 |
| Notes payable | |
$ | 1,646,616 | | |
$ | 1,646,616 | |
Note G –
Related Party Transactions
Certain sales
are through contracts entered into Kurve Therapeutics, Inc., a related party through common ownership. The Company manufacturers and
distributes devices for these customer, and Kurve Therapeutics receives a 35% commission. During the years ended December 31, 2021 and
2020, the Company recognized revenue of approximately $131,000 and $73,000, respectively, associated with these contracts. As of December
31, 2021, there was a net of $18,395 due from Kurve Therapeutics. As of December 31, 2020, there was a net of $36,905 due to Kurve Therapeutics.
Kurve Technology,
Inc.
Notes to the
Financial Statements
December 31,
2021 and 2020
Note G –
Related Party Transactions (continued)
The Company has
entered into an agreement to sell its assets to a related entity through common ownership, which is designed to retire all Company debt.
While the Company believes in the viability of this transaction, there can be no assurances to that effect.
Note H –
Income Taxes
No provision
or benefit for federal or state income taxes has been reflected for the years ended December 31, 2021 and 2020 since the Company reported
losses and has established a valuation allowance against the total net deferred tax asset.
Significant components
of the Company’s deferred tax assets (liabilities) as of December 31:
| | |
2021 | |
2020 |
| Net operating loss carryforward | |
$ | 1,448,022 | | |
$ | 1,378,954 | |
| Valuation allowance | |
| (1,448,022 | ) | |
| (1,378,954 | ) |
| Net deferred tax asset | |
$ | — | | |
$ | — | |
The entire balance
of the deferred tax assets has been offset by a valuation allowance since the utilization of net operating loss carryforwards cannot
be sufficiently assured at December 31, 2021 and 2020.
At December 31,
2021 and 2020, the Company had federal net operating loss carryforwards of approximately $6,460,000 and $6,150,000, respectively. Approximately
$5,749,000 of the federal net operating loss will expire between 2023 and 2037. Net operating loss beginning in 2018 may be carried forward
indefinitely but are limited to 80% of the taxable income in any one tax period. The utilization of the net operating loss carryforwards
may be limited in the case of certain events including significant changes in ownership interests. The Company’s federal and state
blended tax rate was 22.4% for the years ended December 31, 2021 and 2020.
Note I –
Common Stock Warrants
The following
common stock warrants were outstanding as of December 31, 2021 and 2020:
| Exercise
Price |
Number
Outstanding |
Expiration
Date |
Number
Exercisable |
| $ | 0.64 |
150,000 | |
December
2020 |
150,000 | |
| $ | 1.00 |
3,899,000 | |
2022-2030 |
3,899,000 | |
| | Total
outstanding December 31, 2020 |
4,049,000 | |
|
4,049,000 | |
| | Forfeited |
(150,000) | |
|
(150,000) | |
| $ | 1.00 |
3,899,000 | |
2022-2030 |
3,899,000 | |
| | Total
outstanding December
31, 2021 |
3,899,000 | |
|
3,899,000 | |
Note J –
Stock Options
The following
common stock options were outstanding as of December 31, 2021 and 2020:
| Exercise Price | |
Number Outstanding | |
Expiration Date | |
Number Exercisable |
| $ | 1.00 | | |
| 100,000 | | |
November 30,
2024 | |
| 100,000 | |
| | Total outstanding December 31, 2020 | | |
| 100,000 | | |
| |
| 100,000 | |
| $ | 1.00 | | |
| 100,000 | | |
November 30,
2024 | |
| 100,000 | |
| | Total outstanding December 31, 2021 | | |
| 100,000 | | |
| |
| 100,000 | |
Note K –
Commitments & Contingencies
Operating
Lease
The Company leases
its facilities on a month-to-month basis. Total rent expense for the years ended December 31, 2021 and 2020 was approximately $25,000
and $27,000, respectively.
COVID-19
Due to the COVID-19
outbreak in 2020, the Company was not closed at any point. Additionally, there were no staff furloughed or terminated due to COVID-19.
Overall, the Company was not significantly affected by COVID-19.
Due to the level
of risk this virus may have on the global economy, it is at least reasonably possible that it could have an impact on the operations
of the Company in the near term that could materially impact the Company’s financial statements, however, management does not believe
there will be any future impact. In addition, with vaccines and Technology coming to market, these will also help to mitigate any potential
future losses.
Note L –
Subsequent Events
In preparing
the accompanying financial statements, the Company has evaluated events and transactions for potential recognition or disclosure through
February 11, 2022, the date the financial statements were available to be issued. Management has determined that all subsequent events
have been properly disclosed.
PART III - EXHIBITS
Exhibit Index
Exhibit 2A Amended and Restated Certificate of Incorporation for
Kurve Therapeutics, Inc.
Exhibit 2B: Bylaws
Exhibit 4. Subscription Agreement
Exhibit 6. Convertible Notes
Exhibit 8. Escrow Agreement
Exhibit 11. Written Expert Consent Letter of Assurance Dimensions
Exhibit 12. Legal Opinion of Trae O’Neil High
Exhibit 13: Testing the Waters Materials
Exhibit 16A: Receivership Order
Exhibit 16B: Certificate of Merger and Plan of Merger
SIGNATURE PAGE
Pursuant to the requirements of Regulation A, the issuer certifies
that it has reasonable grounds to believe that it meets all of the requirements for filing on Form 1-A and has duly caused this
Offering Statement to be signed on its behalf by the undersigned, thereunto duly authorized, in the City of Lynnwood, WA, on
May 20, 2022.
(Exact name of the Issuer as specified in its Charter)
Kurve Therapeutics, Inc.
16825 48th Ave West, Suite 434
Lynnwood, WA 98037
(425) 640-9249
By:
s/Marc Giroux
Director, CEO of Kurve Therapeutics, Inc.
(Date): May 20, 2022
Location Signed: Lynnwood, WA
s/Tom McDowell
Director, COO of Kurve Therapeutics, Inc.
(Date): May 20, 2022
Location Signed: Lynnwood, WA
This Offering Statement has been signed by the following Officers
in the capacities and on the dates indicated.
s/Marc Giroux
CEO of Kurve Therapeutics, Inc.
(Date): May 20, 2022
Location Signed: Lynnwood, WA
s/Tom McDowell
COO of Kurve Therapeutics, Inc.
(Date): May 20, 2022
Location Signed: Lynnwood, WA
This Offering Statement has been signed by the following Directors
in the capacities and on the dates indicated.
s/Marc Giroux
Director of Kurve Therapeutics, Inc.
(Date): May 20, 2022
Location Signed: Lynnwood, WA
s/Tom McDowell
Director of Kurve Therapeutics, Inc.
(Date): May 20, 2022
Location Signed: Lynnwood, WA

 7714168
8100 Authentication: 202492909
7714168
8100 Authentication: 202492909